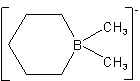
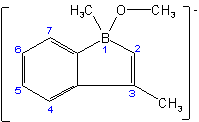
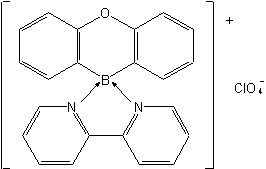
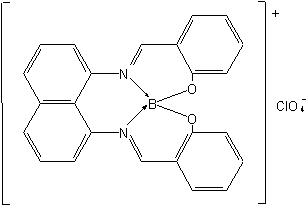
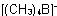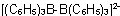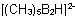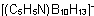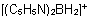Examples to Rule D-7.62
Tetramethylborate(1-)
Hexaphenyldibarate(2-)
Hydropentamethyldiborate(2-)
Tridecahydro(pyridine)decaborate(1-)
Trifluoro[(triphenylphosphonio)-methyl]borate
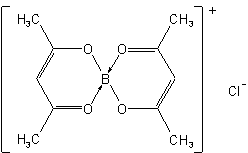
Dihydrobis(pyridine)boron(1+)Bis(2,4-pentanedionato)boron(1+) chloride
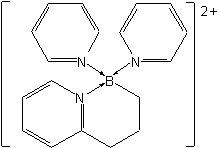
Bis(pyridine)[3-(2-pyridyl)propyl]boron(2+)
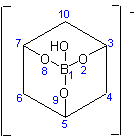
[1,3,5-Cyclohexanetriolato(3-)]hydroxoborate(1-)
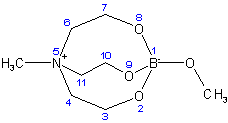
Methoxo[methyltris(2-oxidoethyl)ammonium-O,O,O]borate