

ACD/Name (Chemist Version) offers a standardized set of features for quick and simple generation of IUPAC names, and structures from names. It is a streamlined version of our popular ACD/Name software.
View a full description and pricing on our web store.


The name "methylene" is retained for the radical 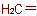
4.2 - The names of bivalent radicals derived from normal alkanes by removal of a hydrogen atom from each of the two terminal carbon atoms of the chain are ethylene, trimethylene, tetramethylene, etc.Examples to Rule A-4.1
Methylidyne Ethylidene Ethylidyne Vinylidene Isopropylidene
Names of the substituted bivalent radicals are derived in accordance with Rules A-2.2 and A-2.25.Examples to Rule A-4.2
Pentamethylene Hexamethylene
The following name is retained:Example to Rule A-4.2a
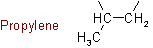
4.3 - Bivalent radicals similarly derived from unbranched alkenes, alkadienes, alkynes, etc., by removing a hydrogen atom from each of the terminal carbon atoms are named by replacing the endings "-ene", "-diene", "-yne", etc., of the hydrocarbon name by "-enylene", "-dienylene", "-ynylene", etc., the positions of the double and triple bonds being indicated where necessary.
The following name is retained:Example to Rule A-4.3
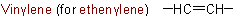
Names of the substituted bivalent radicals are derived in accordance with Rule A-3.4.
4.4 - Trivalent, quadrivalent and higher-valent acyclic hydrocarbon radicals of two or more carbon atoms with the free valences at each end of a chain are named by adding to the hydrocarbon name the terminations "-yl" for a single free valence, "-ylidene" for a double, and "-ylidyne" for a triple free valence on the same atom (the final "e" in the name of the hydrocarbon is elided when followed by a suffix beginning with "-yl"). If different types are present in the same radical, they are cited and numbered in the order "-yl", "-ylidene", "-ylidyne".Example to Rule A-4.3
4.5 - Multivalent radicals containing three or more carbon atoms with free valences at each end of a chain and additional free valences at intermediate carbon atoms are named by adding the endings "-triyl", "-tetrayl", "-diylidene", "diyl-ylidene", etc., to the hydrocarbon name.Examples to Rule A-4.4
Butanediylidene Butanediylidyne 1-Propanyl-3-ylidene Propadienediylidene 2-Pentenediylidyne 1-Butanyliden-4-ylidyne
See Recommendations'93 R-2.5Examples to Rule A-4.5


Published with permission of the IUPAC by Advanced Chemistry Development, Inc., www.acdlabs.com, +1(416)368-3435 tel, +1(416)368-5596 fax. For comments or suggestions please contact [email protected]