










ACD/Name (Chemist Version) offers a standardized set of features for quick and simple generation of IUPAC names, and structures from names. It is a streamlined version of our popular ACD/Name software.
View a full description and pricing on our web store.
 |
||
| Ring size | Unsaturated |
Saturated |
 |
||
| 3 | irene |
irane |
| 4 | ete | etane |
| 5 | ole | olane |
6A |
ine | ane |
6B |
ine | inane |
6C |
inine | inane |
| 7 | epine | epane |
| 8 | ocine | ocane |
| 9 | onine | onane |
| 10 | ecine | ecane |
Note 2 Although a terminal "e" is used on all stems in these recommendations, it is optional. (Stems without the terminal "e" for unsaturated nonnitrogenous rings with six or more ring members are used in CAS index nomenclature, for example, dioxin, and dioxaphosphepin).
Note 3 The stems for ring sizes 3, 4, 7, 8, 9, and 10 may be considered to be derived from numerical prefixes as follows: "ir" from tri, "et" from tetra, "ep" from hepta, "oc" from octa, "on" from nona, and "ec" from deca.
Note 4 The stems "etine" and "oline", which would be consistent with the other stems for unsaturated rings, cannot be used because they were formerly used to name nitrogenous four- and five-membered rings, respectively, having only one of two possible double bonds.
Note 5 Oxine must not be used for pyran because it has been used as a trivial name for quinolin-8-ol.
Note 6 Azine must not be used for pyridine because of its long-established use as a class name for 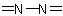 compounds (see R-5.6.6.3).
compounds (see R-5.6.6.3).


Published with permission of the IUPAC by Advanced Chemistry Development, Inc., www.acdlabs.com, +1(416)368-3435 tel, +1(416)368-5596 fax. For comments or suggestions please contact [email protected]