
ACD/Name (Chemist Version) offers a standardized set of features for quick and simple generation of IUPAC names, and structures from names. It is a streamlined version of our popular ACD/Name software.
View a full description and pricing on our web store.

(a) by citing the name of the substituent group R, preceded by the numerical prefix "di-" or "tri-", respectively, as a prefix to the name of the parent hydride azane;
(b) by citing the name of the substituent group R, preceded by "di-" or "tri-", as appropriate, and followed directly, without a space, by the name "amine" .
.
Unsymmetrical secondary and tertiary amines,Examples to R-5.4.2
(C6H5)2NH(a) Diphenylazane
(b) Diphenylamine
(C2H5)3N
(a) Triethylazane
(b) Triethylamine
(ClCH2-CH2)2NH
(a) Bis(2-chloroethyl)azane
(b) Bis(2-chloroethyl)amine
2,2'-Dichlorodiethylamine
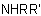 ,
, 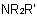 , and
, and 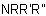 , may be named according to three methods as follows:
, may be named according to three methods as follows:(a) as substituted derivatives of the parent hydride azane;
(b) as N-substituted derivatives of a primary amine RNH2 or a secondary amine R2NH;
(c) by citing the names of all substituent groups, R, 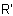 , or R", preceded by appropriate numerical prefixes, and followed directly, without a space, by the class name "amine".
, or R", preceded by appropriate numerical prefixes, and followed directly, without a space, by the class name "amine".
Substituent groups in names of unsymmetrical secondary and tertiary amines are ordered alphabetically.
Examples to R-5.4.2



Published with permission of the IUPAC by Advanced Chemistry Development, Inc., www.acdlabs.com, +1(416)368-3435 tel, +1(416)368-5596 fax. For comments or suggestions please contact [email protected]