
ACD/Name (Chemist Version) offers a standardized set of features for quick and simple generation of IUPAC names, and structures from names. It is a streamlined version of our popular ACD/Name software.
View a full description and pricing on our web store.

 .
.
R-5.7.7.1 Symmetrical anhydrides. Symmetrical anhydrides of monobasic acids, substituted or unsubstituted, are named by replacing the term "acid"
of the acid name by the class name "anhydride" .
.
Cyclic anhydrides formed from two acid groups attached to the same parent hydride structure may be named in the same manner as symmetrical anhydrides, or as heterocyclic compounds.Examples to R-5.7.7.1
R-5.7.7.2 Unsymmetrical (mixed) anhydrides. Anhydrides derived from different monobasic acids are named by citing the first parts of the names of the two acids (i.e., the parts preceding the term acid) in alphbetical order, followed by the class name "anhydride" as a separate word.Examples to R-5.7.7.1
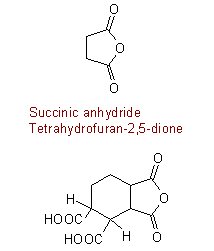
Cyclohexane-1,2,3,4-tetracarboxylic acid 3,4-anhydride
1,3-Dioxooctahydro-2-benzofuran-4,5-dicarboxylic acid
1,3-Dioxooctahydrobenzo[c]furan-4,5-dicarboxylic acid
1,3-Dioxooctahydroisobenzofuran-4,5-dicarboxylic acid
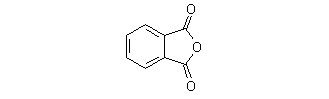
Phthalic anhydride
1,3-Dihydro-2-benzofuran-1,3-dione
1,3-Dihydrobenzo[c]furan-1,3-dione
1,3-Dihydroisobenzofuran-1,3-dione
R-5.7.7.3 Chalcogen analogues of anhydrides having the general structureExamples to R-5.7.7.2
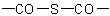 ,
, 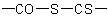 , or
, or 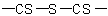 are named in
the same way as their oxygen analogues, using the class name "thioanhydride". The sulfur linkage between the acyl groups is given by the replacement prefix "thio-"; other sulfur atoms are indicated
by "thio-" prefixes cited with the appropriate acyl group.
are named in
the same way as their oxygen analogues, using the class name "thioanhydride". The sulfur linkage between the acyl groups is given by the replacement prefix "thio-"; other sulfur atoms are indicated
by "thio-" prefixes cited with the appropriate acyl group.
Alternatively, such thioanhydrides may be named on the basis of the parent hydride name "sulfane", the corresponding class name being "diacylsulfanes".
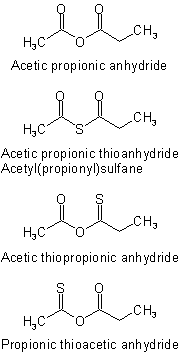
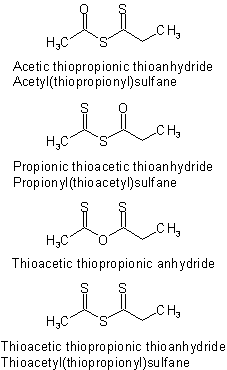
The various unsymmetrical thioanhydrides derived from acetic propionic anhydride are named as follows:Examples to R-5.7.7.3



Published with permission of the IUPAC by Advanced Chemistry Development, Inc., www.acdlabs.com, +1(416)368-3435 tel, +1(416)368-5596 fax. For comments or suggestions please contact [email protected]