November 11, 2021
by Mikhail Elyashberg, Leading Researcher, ACD/Labs
Iridiod Glycoside
Clerodendrum (Lamiaceae) is a genus composed of 580 species. It is found in tropical and subtropical regions and mainly includes small trees, shrubs, and herbs. Many members of the genus havebeen used in traditional medicine around the world.
C. myricoides is a shrub with bluish flowers that has been used extensively in traditional medicine. Earlier investigations led to the isolation of spermidine alkaloids, phenolic glycosides, and cyclopeptides. Erdelyi and coworkers [1] have carried out a phytochemical investigation of its root extract. In their article, the authors reported the isolation and characterization of a new iridoid glycoside 1 from the methanol root extract.
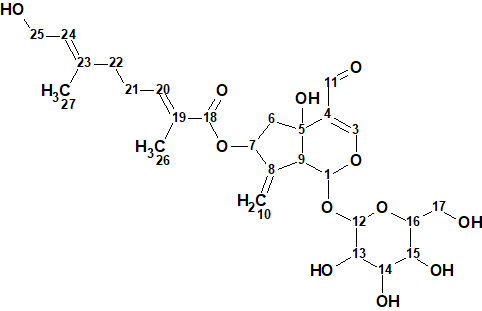
1
Compound 1 was isolated as an amorphous white powder, and was assigned the molecular formula C26H36O12 based on HR-ESI-MS ([M + H]+ m/z 541.2271, calcd 541.2278) and NMR data analysis. The NMR spectroscopic data reported by authors are presented in Table 1.
Table 1. NMR spectroscopic data used for structure elucidation of compound 1.[1]
| Label | δC | δC calc (HOSE) | CHn | δH | COSY | H to C HMBC |
| C 1 | 97.1 | 95.72 | CH | 6.05 | C 5, C 12, C 3 | |
| C 3 | 165 | 163.13 | CH | 7.44 | C 5, C 1, C 4, C 11 | |
| C 4 | 123.6 | 122.8 | C | |||
| C 5 | 69.9 | 68.96 | C | |||
| C 6 | 42.6 | 43.13 | CH2 | 3.08 | C 9, C 5, C 7, C 4, C 8 |
|
| C 6 | 42.6 | 43.13 | CH2 | 2.02 | ||
| C 7 | 74 | 73.37 | CH | 5.25 | ||
| C 8 | 146.8 | 145.34 | C | |||
| C 9 | 52.9 | 52 | CH | 3.18 | C 5, C 7, C 8 | |
| C 10 | 114.3 | 114.07 | CH2 | 5.29 | C 9, C 7 | |
| C 10 | 114.3 | 114.07 | CH2 | 5.38 | C 27, C 22 | |
| C 11 | 192.4 | 190.65 | CH | 9.3 | C 5, C 4, C 3 | |
| C 12 | 100.4 | 99.48 | CH | 4.64 | 3.2 | C 1 |
| C 13 | 74.4 | 72.87 | CH | 3.2 | 3.39, 4.64 | C 14, C 12 |
| C 14 | 77.5 | 78.01 | CH | 3.39 | 3.20, 3.27 | C 16 |
| C 15 | 71.6 | 70.57 | CH | 3.27 | 3.37, 3.39 | C 14 |
| C 16 | 78.6 | 76.54 | CH | 3.37 | 3.27, 3.94 | C 15 |
| C 17 | 62.8 | 61.49 | CH2 | 3.94 | 3.37 | |
| C 17 | 62.8 | 61.49 | CH2 | 3.67 | C 16 | |
| C 18 | 169.2 | 166.86 | C | |||
| C 19 | 128.7 | 129.8 | C | |||
| C 20 | 143.9 | 143.26 | CH | 6.79 | C 26, C 18 | |
| C 21 | 28 | 27.6 | CH2 | 2.35 | C 22, C 19, C 23, C 20 |
|
| C 22 | 39 | 39.2 | CH2 | 2.15 | C 27, C 21, C 24, C 23, C 20 |
|
| C 23 | 138.4 | 138.4 | C | |||
| C 24 | 125.7 | 125.7 | CH | 5.38 | ||
| C 25 | 59.3 | 59.4 | CH2 | 4.08 | C 24, C 23 | |
| C 26 | 12.5 | 12.39 | CH3 | 1.83 | C 19, C 20, C 18 | |
| C 27 | 16.2 | 16.2 | CH3 | 1.69 | C 22, C 24, C 23 |
The molecular formula and spectroscopic data were entered in ACD/Structure Elucidator (ACD/SE) to challenge the program. The molecular Connectivity Diagram (MCD) automatically created by the program is shown in Figure 1.

Figure 1. Molecular connectivity diagram. Hybridizations of carbon atoms are marked by corresponding colors: sp2—violet, sp3—blue, not sp (sp2 or sp3)—light blue. Labels “ob” and “fb” are set by the program to carbon atoms for which neighboring with heteroatom is either obligatory (ob) or forbidden (fb). HMBC connectivities are marked by green arrows, while COSY connectivities—by blue arrows.
No manual edits of the MCD were done, and structure generation was initiated. It was completed with the following results: k = 4080→ (spectral filtering)→1555→ (removal of duplicates)→ 1042, tg =1min 18 sec. After 13C chemical shift prediction for structures included into the output file using the three methods available in ACD/SE (incremental approach, neural networks and HOSE-code based technique), the structures were ranked in increasing order of values dA – average deviations between experimental and calculated chemical shifts. The four top ranked structures are shown in Figure 2.
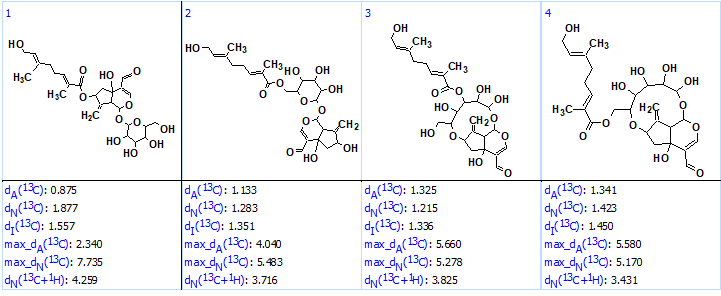
Figure 2. The top four ranked structures of the output file. Average deviations of 13C chemical shifts determined by the three methods are denoted as dA (HOSE code-based), dN (neural networks) and dI (increments).
Structure #1 coincided with the structure of compound 1, but the average deviations calculated for the remaining structures are in the limits of accuracy provided by empirical methods of 13C chemical shift prediction. A visual analysis of the ranked file as well as the MCD clearly showed that the correct structure must contain a glycoside fragment (structure 2):
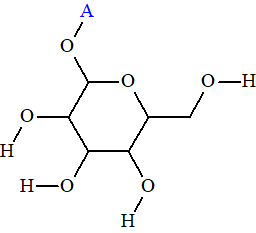
2
Therefore, the structures containing this fragment were selected from the output file (Tools/Check Structures by/User GoodList) and ranked again (see Figure 3).
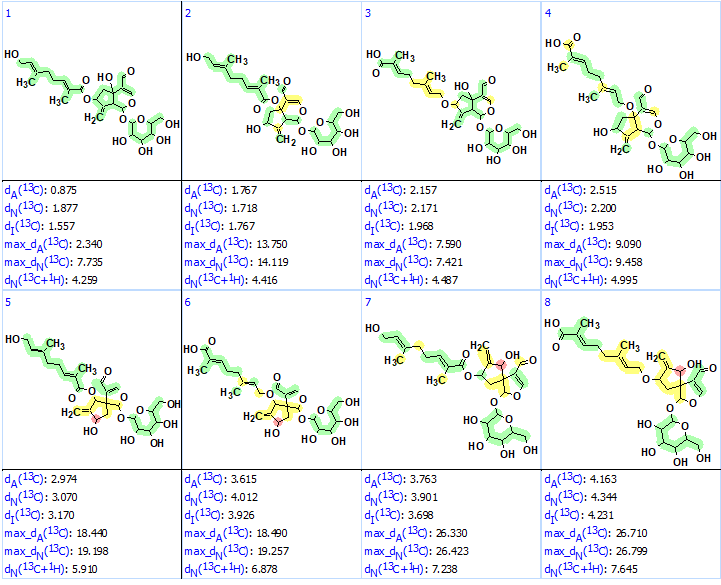
Figure 3. The eight structures of the output file containing substructure 2. Each atom is colored to mark a difference between its experimental and calculated 13C chemical shifts. The green color represents a difference between 0 to 3 ppm, yellow was >3 to 15 ppm, and red > 15 ppm.
We see that presence of fragment 2 in all structures is confirmed by the chemical shift prediction—all carbon atoms of glycoside are green. All carbon atoms of structure #1 are also green. As the dA values of structures #1–#4 are within the range common for a correct structure determined by ACD/SE, DP4 probabilities were calculated for these structures (see Figure 4).
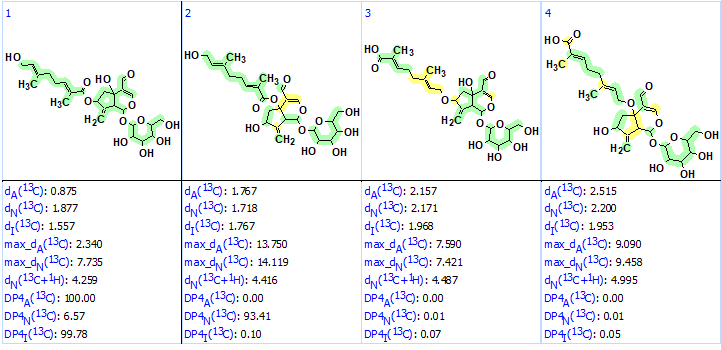
Figure 4. DP4 probabilities calculated for the first four structures.
We see that not only values of average deviations but DP4 probabilities are also confirming the validity of structure #1. Because dN(#2) – dN(#1) = 0.16 the neural networks distinguished structure #2 as the most probable (DP4N=93), but the two remaining approaches have “voted” for structure #1. Moreover we see that the maximum deviations found by both Neural Networks and HOSE-codes based predictions for structure #2 are at least twice as large as for the ones for structure #1, further supporting the validity of structure #1.
The elucidated structure together with the assigned 13C chemical shifts is presented below:
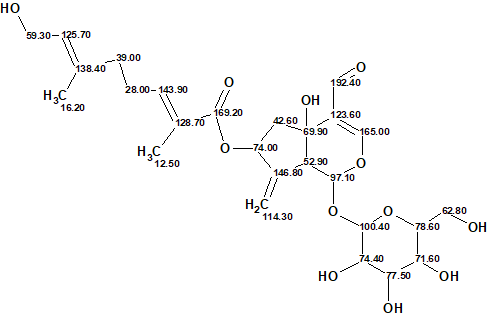
Thus, the correct structure of the irioid glycoside 1 was reliably determined using ACD/SE in a fully automatic mode.
References
- D. Umereweneza, J.T. Molel, J. Said, Y. Atilaw, T. Muhizi, E. Trybala, T. Bergstrom, A. Gogoll, M. Erdelyi. Antiviral iridoid glycosides from Clerodendrum myricoides, Fitoterapia 155 (2021) 105055


