August 1, 2020
by Mikhail Elyashberg, Leading Researcher, ACD/Labs
Leonuketal
Leonurus Japonicus is a plant that is used in common traditional Chinese medicine. In fact, the aerial parts of it are in the Chinese Pharmacopoeia. Many diterpene metabolites have been discovered on plants of the same genus, with the common characteristics of the furanolabdane class. This class has been found to have interesting bioactivities and so research on plants of this species continues.
Xiong et al [1] investigated the bioactive secondary metabolites from L. japonicus. As a result, a diterpenoid with an unprecedented tetracyclic skeleton, namely leonuketal (1), was discovered. The highlighted structural feature includes a bridged spiroketal moiety fused with a ketal-γ-lactone ring.
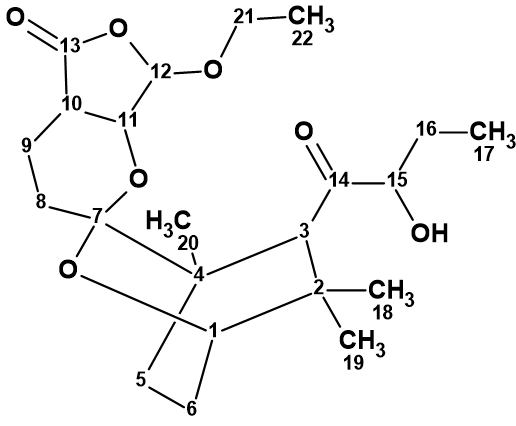
1
Leonuketal (1) was obtained as a colorless gum with the molecular formula C22H34O7 determined by HR-ESI-MS (m/z 433.2194 [M + Na]+, calcd 433.2202), which indicates six degrees of unsaturation. The IR spectrum showed characteristic absorption bands for hydroxyl (3362 cm−1) and carbonyl (1787 and 1726 cm−1) groups.
1D and HSQC spectroscopic data as well as the key HMBC 2D NMR correlations which were utilized for structure elucidation of leonuketal are presented in Table 1.
Table 1. 1D and 2D NMR spectroscopic data of Leonuketal.
| Label | δC | δC calc (HOSE) | CHn | δH | H to C HMBC |
| C 1 | 78.4 | 79.45 | CH | 3.46 | C 7 |
| C 2 | 37.3 | 36.87 | C | ||
| C 3 | 52.8 | 52.57 | CH | 2.94 | C 5, C 2, C 4, C 1, C 15, C 7, C 14 |
| C 4 | 39.6 | 45.48 | C | ||
| C 5 | 23.2 | 40.61 | CH2 | 2.39 | |
| C 5 | 23.2 | 40.61 | CH2 | 1.56 | |
| C 6 | 26.3 | 18.87 | CH2 | 1.81 | |
| C 6 | 26.3 | 18.87 | CH2 | 1.87 | |
| C 7 | 100.7 | 103.21 | C | ||
| C 8 | 26.3 | 28.39 | CH2 | 1.79 | |
| C 8 | 26.3 | 28.39 | CH2 | 1.34 | C 4 |
| C 9 | 16.4 | 18.95 | CH2 | 2.24 | |
| C 9 | 16.4 | 18.95 | CH2 | 2 | C 8, C 7, C 13 |
| C 10 | 37.5 | 41.32 | CH | 2.78 | |
| C 11 | 73.2 | 72.22 | CH | 4.48 | C 9, C 7, C 13 |
| C 12 | 105.9 | 103.87 | CH | 5.31 | C 10, C 21, C 11, C 13 |
| C 13 | 177.3 | 175.5 | C | ||
| C 14 | 213.8 | 216.21 | C | ||
| C 15 | 80.1 | 79.14 | CH | 3.96 | |
| C 16 | 27.2 | 25.32 | CH2 | 1.9 | C 14 |
| C 16 | 27.2 | 25.32 | CH2 | 1.4 | |
| C 17 | 10.2 | 9.06 | CH3 | 0.98 | C 16, C 15 |
| C 18 | 23.7 | 19.1 | CH3 | 0.81 | C 2, C 3, C 1 |
| C 19 | 30.1 | 25.68 | CH3 | 1.24 | C 2, C 3, C 1 |
| C 20 | 19.1 | 21.31 | CH3 | 0.75 | C 5, C 4, C 3, C 7 |
| C 21 | 65.6 | 68.83 | CH2 | 3.81 | |
| C 21 | 65.6 | 68.83 | CH2 | 3.69 | |
| C 22 | 15.3 | 14.88 | CH3 | 1.2 |
We used these data for challenging ACD/Structure Elucidator (ACD/SE). In so doing, we entered the 13C, 1H , HSQC and HMBC data into the program. The Molecular Connectivity Diagram (MCD) produced by the software is shown in Figure 1.
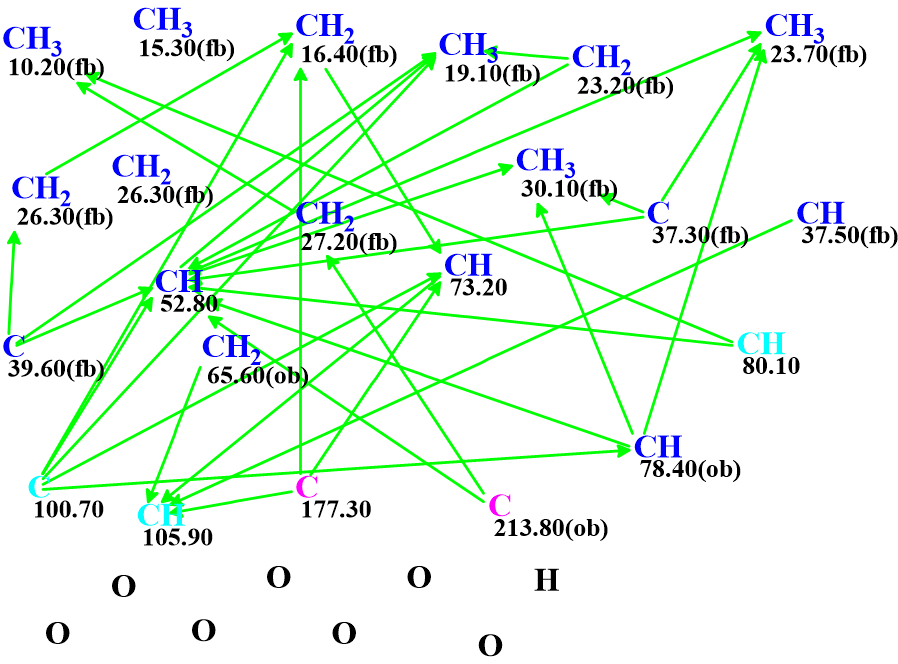
Figure 1. Molecular connectivity diagram.
The carbon atom hybridization was automatically assigned for all atoms except C 100.7, C 105.9 and C 80.1. The latter are marked by light blue color, which indicates that their hybridization is ambiguous – not sp, that is sp2 or sp3.
Structure generation was initiated from the MCD, which was completed with the following results: k= 6, tg = 1 s.
13C chemical shift prediction using three empirical methods (incremental, neural networks and HOSE code based) was carried out for structures of the output file and structures were ranked in increasing order of average deviations of calculated spectra from the experimental one. The ranked output file is shown in Figure 2.
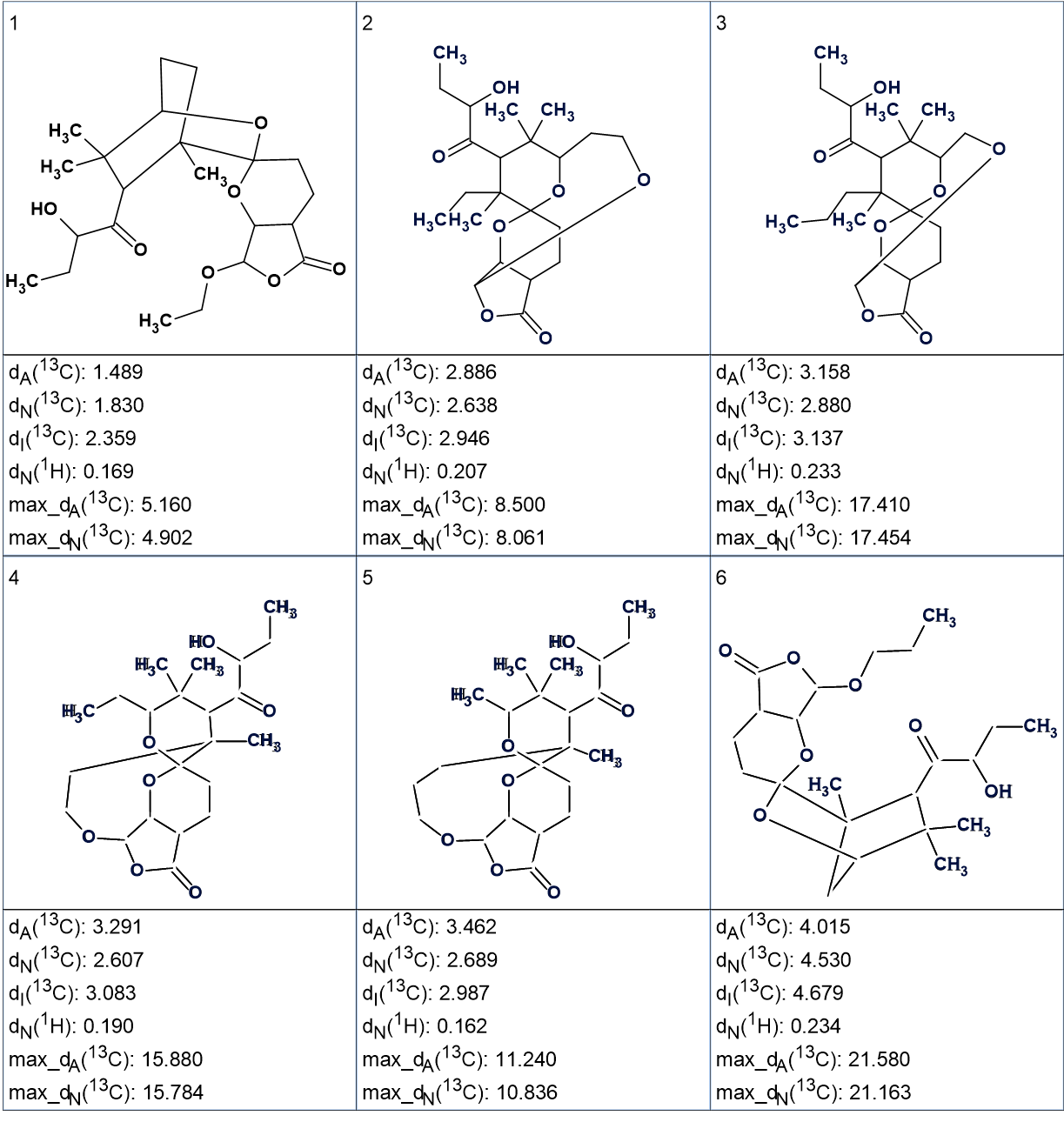
Figure 2
We see that the first ranked structure coincides with structure of leonuketal determined by the authors [1]. The structure of leonuketal with automatically assigned 13C chemical shifts is shown below.
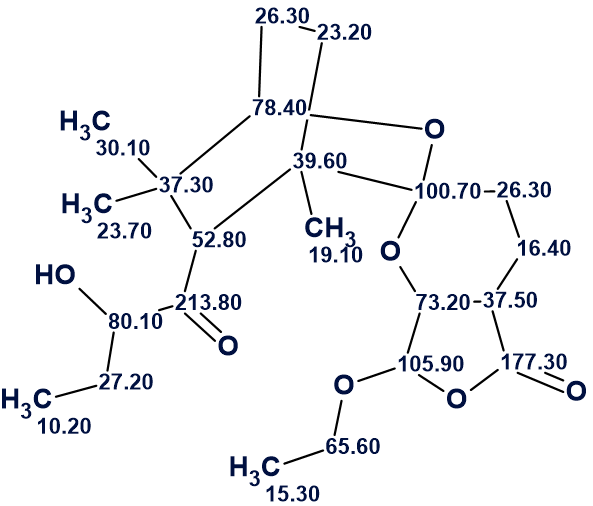
Thus the structure of a new natural product characterized by an unprecedented skeleton was elucidated fully automatically in almost no time.
References
- L. Xiong, Q.-M. Zhou, Y. Zou, M.-H. Chen, L. Guo, G.-Y. Hu, Z.-H. Liu, C. Peng. (2015). Leonuketal, a Spiroketal Diterpenoid from Leonurus japonicus. Org. Lett., 17(24): 6238–6241, DOI: 10.1021/acs.orglett.5b03227


