
ACD/Name (Chemist Version) offers a standardized set of features for quick and simple generation of IUPAC names, and structures from names. It is a streamlined version of our popular ACD/Name software.
View a full description and pricing on our web store.

Exceptions:Examples to Rule A-23.1
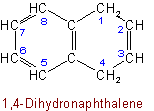
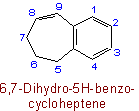
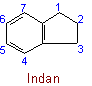
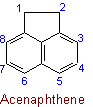
The following names are retained:
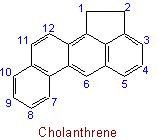
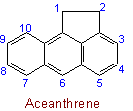
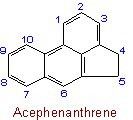
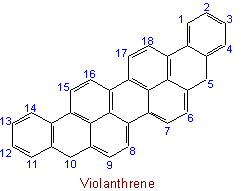
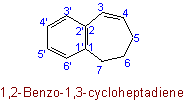
23.2 - When there is a choice, the carbon atoms to which hydrogen atoms are added are numbered as low as possible.
23.3 - Substituted polycyclic hydrocarbons are named according to the same principles as substituted monocyclic hydrocarbons (see Rules A-12 and A-61).Examples to Rule A-23.2
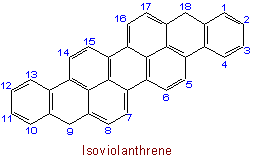
23.5 (Alternate to part of Rule A-23.1) - The names of "ortho-fused" polycyclic hydrocarbons which have (a) less than the maximum number of non-cumulative double bonds, (b) at least one terminal unit which is most conveniently named as an unsaturated cycloalkane derivative, and (c) a double bond at the positions where rings are fused together, may be derived by joining the name of the terminal unit to that of the other component by means of a letter "o" with elision of a terminal "e". The abbreviations for fused aromatic systems laid down in Rule A-21.4 are used, and the exceptions of Rule A-23.1 apply.
See Recommendations'93 R-2.4.1, R-9.1, Tables 20 and 20(a)Examples to Rule A-23.5



Published with permission of the IUPAC by Advanced Chemistry Development, Inc., www.acdlabs.com, +1(416)368-3435 tel, +1(416)368-5596 fax. For comments or suggestions please contact webmaster@acdlabs.com