
ACD/Name (Chemist Version) offers a standardized set of features for quick and simple generation of IUPAC names, and structures from names. It is a streamlined version of our popular ACD/Name software.
View a full description and pricing on our web store.

72.2 - Names of symmetrical compound radicals are formed by juxtaposing the names of the individual radicals starting with the central radical e.g., methylenedioxy, oxydiethylene; or the trivial name, if any, for the compound radical may be used.Examples to Rule C-72.1
Note: On this nomenclature system, unsymmetrical compound radicals are not used for linking identical units because of difficulty in assigning unambiguous numbering to the complete structure. Instead, simple substitutive nomenclature is used, e.g.:Examples to Rule C-72.2
(In the last example the compound radical name is formed by starting with the central radical "oxy-", followed by "-bis-", and adding successively the names of the radicals "ethylene" (-CH2-CH2-) and "nitrilo" (-N=), and finally the name of the unit "propionic acid" is added.)
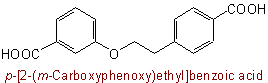
See Recommendations'93 R-2.4.4 and R-1.2.8


Published with permission of the IUPAC by Advanced Chemistry Development, Inc., www.acdlabs.com, +1(416)368-3435 tel, +1(416)368-5596 fax. For comments or suggestions please contact [email protected]