
ACD/Name (Chemist Version) offers a standardized set of features for quick and simple generation of IUPAC names, and structures from names. It is a streamlined version of our popular ACD/Name software.
View a full description and pricing on our web store.

Exceptions: Names of the type benzenediazonium (Rule C-931), uronium (Rule C-973), and thiouronium (Rule C-974) are used as laid down in the Rules cited.Examples to Rule C-82.1
Tetramethylammonium Dimethyloxonium Diphenyliodonium Ethylenebromonium
82.2 - If the positive charge of an organic cation is derived by fixation of a proton on a hetero atom of a parent compound whose name does not end in amine, the name of the ion is formed by adding "-ium" to the name of the organic parent compound, with elision of a terminal "e" (if present). When there is more than one hetero atom able to fix the proton, the point of attachment of the proton may be shown by the "indicated hydrogen" method.
82.3 - When replacement ("a") nomenclature is used (see Rule B-6) the ending "-a" of an affix oxa, thia, aza, etc., is changed to "-onia".Examples to Rule C-82.2
Anilinium Guanidinium 2-Phenylhydrazinium 1-Methylhydrazinium
(for numbering see Rule C-921.5)Acetonium 1,4-Dioxanium 1-Methylpyridinium Imidazolium Glycinium Prolinium 1,4-Dithian-2-iminium 9aH-Quinolizinium
82.4 - An organic cation formed by addition of a proton to an unsaturated hydrocarbon is named by adding "-ium" to the name of the hydrocarbon, with elision of terminal "e" if present. If desired the point of attachment of the proton is shown by the "indicated hydrogen" method. If aExample to Rule C-82.3
 -complex is to be specified, the prefix
-complex is to be specified, the prefix 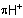 is used. If the position of the charge is to
be specified, the appropriate locant is placed before the ending "-ium".
is used. If the position of the charge is to
be specified, the appropriate locant is placed before the ending "-ium".
See Recommendations'93 R-5.8.2Examples to Rule C-82.4
Ethenium Benzenium Allenium
(position ofunknown)
Heptamethylbenzenium 6H-Azulenium


Published with permission of the IUPAC by Advanced Chemistry Development, Inc., www.acdlabs.com, +1(416)368-3435 tel, +1(416)368-5596 fax. For comments or suggestions please contact [email protected]