ACD/Name (Chemist Version) offers a standardized set of features for quick and simple generation of IUPAC names, and structures from names. It is a streamlined version of our popular ACD/Name software.
View a full description and pricing on our web store.
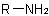 ,
for example, ethylamine, becomes a functional parent compound; the latter method is better illustrated with substituted or unsymmetrical secondary amines (see R-5.4.2). The first method
is used by Beilstein and the second was used in Chemical Abstracts index nomenclature prior to Volume 76 (1972). Both methods are illustrated in the 1979 edition of the IUPAC
Nomenclature of Organic Chemistry
,
for example, ethylamine, becomes a functional parent compound; the latter method is better illustrated with substituted or unsymmetrical secondary amines (see R-5.4.2). The first method
is used by Beilstein and the second was used in Chemical Abstracts index nomenclature prior to Volume 76 (1972). Both methods are illustrated in the 1979 edition of the IUPAC
Nomenclature of Organic Chemistry .
.


Published with permission of the IUPAC by Advanced Chemistry Development, Inc., www.acdlabs.com, +1(416)368-3435 tel, +1(416)368-5596 fax. For comments or suggestions please contact webmaster@acdlabs.com