hydrazino)benzoate

ACD/Name (Chemist Version) offers a standardized set of features for quick and simple generation of IUPAC names, and structures from names. It is a streamlined version of our popular ACD/Name software.
View a full description and pricing on our web store.

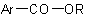 derived from an acyclic alcohol,
derived from an acyclic alcohol, 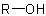 , and an
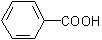
arenecarboxylic acid,
, and an
arenecarboxylic acid, 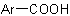 .
.
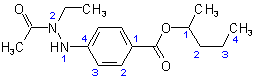
(a) The acyclic alcohol from which this ester is derived is described by the first word '1-methylbutyl'. The prefix 'butyl' indicates a four-carbon chain derived from the parent hydride 'butane'. The prefix 'methyl-' with its locant '1' indicates that a methyl group is at position '1' of the butyl group which, by definition, also bears the free valence, resulting in the following structure and numbering for the alcoholic component.
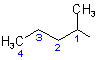
 (I)
(I)
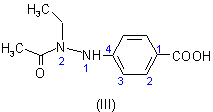
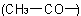 and 'ethyl'
and 'ethyl' 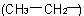 at position 2 on the
parent substituent 'hydrazino',
derived from the parent hydride hydrazine. By definition, the free valence for the hydrazino substituent must have the locant '1'; thus, the structure and numbering of the substituent
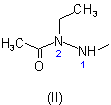
of the acid component is shown as (II). The structure and numbering for the acid component of this ester are given as (III).
at position 2 on the
parent substituent 'hydrazino',
derived from the parent hydride hydrazine. By definition, the free valence for the hydrazino substituent must have the locant '1'; thus, the structure and numbering of the substituent
of the acid component is shown as (II). The structure and numbering for the acid component of this ester are given as (III).



Published with permission of the IUPAC by Advanced Chemistry Development, Inc., www.acdlabs.com, +1(416)368-3435 tel, +1(416)368-5596 fax. For comments or suggestions please contact [email protected]