
ACD/Name (Chemist Version) offers a standardized set of features for quick and simple generation of IUPAC names, and structures from names. It is a streamlined version of our popular ACD/Name software.
View a full description and pricing on our web store.

The appropriate prefix "tricyclo-", "tetracyclo-", etc., is substituted for "bicyclo-" before the name of the open-chain hydrocarbon containing the same total number of carbon atoms. Radicals derived from these hydrocarbons are named according to the principles set forth in Rule A-31.4.
32.12 - A polycyclic system is regarded as containing a number of rings equal to the number of scissions required to convert the system into an open-chain compound.
32.13 - The word "cyclo" is followed by brackets containing, in decreasing order, numbers indicating the number of carbon atoms in:
the two branches of the main ring,
the main bridge,
the secondary bridges.
32.21 - The main ring and the main bridge form a bicyclic system whose numbering is made in compliance with Rule A-31.Examples to Rule A-32.13
32.22 - The location of the other or so-called secondary bridges is shown by superscripts following the number indicating the number of carbon atoms in the said bridges.
32.23 - For the purpose of numbering, the secondary bridges are considered in decreasing order. The numbering of any bridge follows from the part already numbered, proceeding from the
highest-numbered bridge head. If equal bridges are present, the numbering begins at the highest. numbered bridgehead.
32.31 - When there is a choice, the following criteria are considered in turn until a decision is made:
(a) The main ring shall contain as many carbon atoms as possible, two of which must serve as bridgeheads for the main bridge.
Examples to Rule A-32.31(a)
Examples to Rule A-32.31(b)
Examples to Rule A-32.31(c)
(b) The main bridge shall be as large as possible. 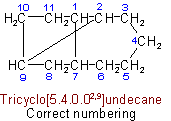
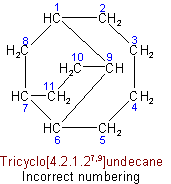
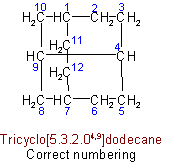
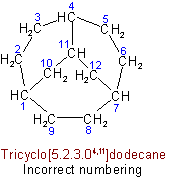
(c) The main ring shall be divided as symmetrically as possible by the main bridge. 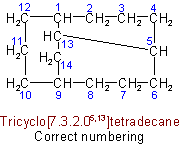
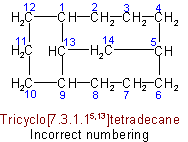
(d) The superscripts locating the other bridges shall be as small as possible (in the sense indicated in Rule A-2.2).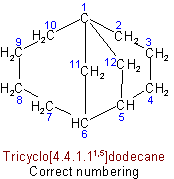
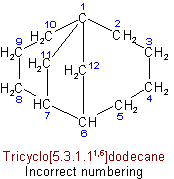
See Recommendations'93 R-2.4.2Examples to Rule A-32.31(d)



Published with permission of the IUPAC by Advanced Chemistry Development, Inc., www.acdlabs.com, +1(416)368-3435 tel, +1(416)368-5596 fax. For comments or suggestions please contact [email protected]