
ACD/Name (Chemist Version) offers a standardized set of features for quick and simple generation of IUPAC names, and structures from names. It is a streamlined version of our popular ACD/Name software.
View a full description and pricing on our web store.

313.2 - Acyclic monoacyl derivatives of cyclic compounds are named (a) by the method of Rule C-313.1, (b) by the radicofunctional method of Rule C-312.2, (c) by prefixing the name of the acyl group to the name of the cyclic component, or (d) if the cyclic component is benzene or naphthalene by changing the ending "-ie acid" or "-oie acid" of the name of the acid corresponding to the acyl group to "-ophenone" or "-onaphthone", respectively.Examples to Rule C-313.1
Exceptions:Examples to Rule C-313.2
The following are examples of trivial names retained for ketones:
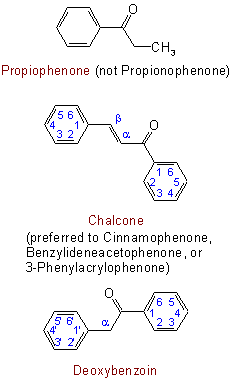
313.3 - When a carbonyl group is attached directly to carbon atoms in two ring systems, the compound is named by the radicofunctional method provided that no other substituent is present having priority for citation as principal group (compare Rule C-316.2).
Exception:Examples to Rule C-313.3
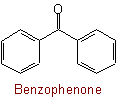
313.4 - The name of a polyketone in which two or more contiguous carbonyl groups have rings attached at each end is formed (a) by the radicofunctional method with the functional class name "diketone", "triketone", etc., or (b) by substitutive nomenclature with "-one" suffixes.
Exceptions:Examples to Rule C-313.4
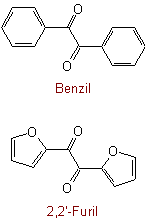
See Recommendations'93 R-5.6.2


Published with permission of the IUPAC by Advanced Chemistry Development, Inc., www.acdlabs.com, +1(416)368-3435 tel, +1(416)368-5596 fax. For comments or suggestions please contact [email protected]