
ACD/Name (Chemist Version) offers a standardized set of features for quick and simple generation of IUPAC names, and structures from names. It is a streamlined version of our popular ACD/Name software.
View a full description and pricing on our web store.

Exception:Examples to Rule C-815.1
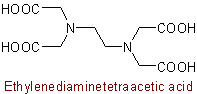
Note: By established custom the above name may be used in place of the systematic name Ethylenedinitrilotetraacetic acid. Extension of the former nomenclature to analogous cases should be restricted to cases where intelligibility is thereby clearly improved; a border line example is:
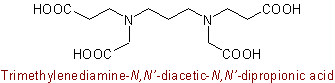
Although these are conjunctive names they do not conform to Rule C-0.5.
815.2 - A group -NH- forming a bridge between two carbon atoms of a ring may be named by a prefix "epimino-" or "imino-" (see Rule B-15.1) .
815.3 - A compound containing a group >C=NH may be named (a) from the corresponding -CH2 compound by means of a suffix "-imine" or, if a group having priority for citation as principal group is also present, by a prefix "imino-", or (b) by citing the name of the bivalent radicalExample to Rule C-815.2
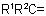 as prefix to "amine''. Compounds
as prefix to "amine''. Compounds 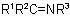 have the class
name "azomethines". When the nitrogen atom is substituted, this class of compound has the generic name "Schiff's bases".
have the class
name "azomethines". When the nitrogen atom is substituted, this class of compound has the generic name "Schiff's bases".
Exception: Quinones from which one or more atoms of quinonoid oxygen have been replaced by =NH or =NR are named by following the name of the quinone with the word "imine", "diimine", etc.; substituents on nitrogen are named as prefixes.Examples to Rule C-815.3
See Recommendations'93 R-5.4Examples to Rule C-815.3


Published with permission of the IUPAC by Advanced Chemistry Development, Inc., www.acdlabs.com, +1(416)368-3435 tel, +1(416)368-5596 fax. For comments or suggestions please contact [email protected]