
ACD/Name (Chemist Version) offers a standardized set of features for quick and simple generation of IUPAC names, and structures from names. It is a streamlined version of our popular ACD/Name software.
View a full description and pricing on our web store.

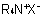 (where the R's may be the same or different), are named by one of the following methods:
(where the R's may be the same or different), are named by one of the following methods:816.1 - The compound is named as a substituted ammonium salt or hydroxide; the names of the substituting radicals precede the word "ammonium" and then the name of the anion is added.
Exceptions:Examples to Rule C-816.1
The following names are retained for the unsubstituted compounds:
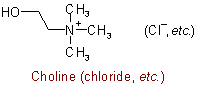
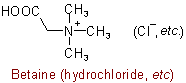
816.2 - When the compound can be considered as derived from a base whose name does not end in "-amine", its quaternary nature is denoted by adding "-ium" to the name of that base, with elision of terminal "e" if present; substituent groups are cited as prefixes, and the name of the anion is added at the end.
816.3 - In complex cases, the prefixes "amino-" and "imino-" may be changed to "ammonio-" and "iminio-" and are followed by the name of the molecule representing the most complex group attached to this nitrogen atom and preceded by the names of the other radicals attached to this nitrogen atom; finally the name of the anion is added (see also Rules C-85 and C-87).Examples to Rule C-816.2
816.4 - When the above rules cannot be applied or lead to inconvenient names, recourse may be had to two traditional methods of naming salts of organic bases, namely: (a) the unaltered name of the base is followed by the name of the anion; and (b), for salts of hydrohalogen acids only, the unaltered name of the base is followed by hydrofluoride, hydrochloride, hydrobromide, or hydriodide, as the case may be. These methods are retained for use also in simpler cases but the procedure of Rule C-816.2 is preferred.Examples to Rule C-816.3
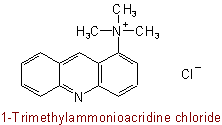
or, by Rule C-816.1, 1-Acridinyltrimethylammonium chloride
Examples, where the structure is indefinite and Rule C-816.2 thus cannot be applied:
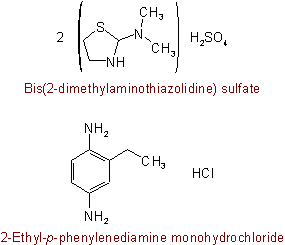
816.5 - Complexes formed from bases and phenols are named by citing the name of the base followed by that of the phenol in its anion form.
See Recommendations'93 R-5.4Examples to Rule C-816.5
Aniline picrate C6H5NH2,C6H3N3O7 Indole styphnate C8H7N,C6H3N3O8


Published with permission of the IUPAC by Advanced Chemistry Development, Inc., www.acdlabs.com, +1(416)368-3435 tel, +1(416)368-5596 fax. For comments or suggestions please contact [email protected]