

ACD/Name (Chemist Version) offers a standardized set of features for quick and simple generation of IUPAC names, and structures from names. It is a streamlined version of our popular ACD/Name software.
View a full description and pricing on our web store.


11.2 - Univalent radicals derived from cycloalkanes (with no side chains) are named by replacing the ending "-ane" of the hydrocarbon name by "-yl", the carbon atom with the free valence being numbered as 1. The generic name of these radicals is "cycloalkyl".Examples to Rule A-11.1
11.3 - The names of unsaturated monocyclic hydrocarbons (with no side chains) are formed by substituting "-ene", "-adiene", "-atriene", "-yne", "-adiyne", etc., for "-ane" in the name of the corresponding cycloalkane. The double and triple bonds are given numbers as low as possible as in Rule A-3.3.Examples to Rule A-11.2
The name "benzene" is retained.Examples to Rule A-11.3
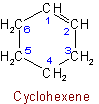
11.4 - The names of univalent radicals derived from unsaturated monocyclic hydrocarbons have the endings "-enyl", "-ynyl", "-dienyl", etc., the positions of the double and triple bonds being indicated according to the principles of Rule A-3.3. The carbon atom with the free valence is numbered as 1, except as stated in the rules for terpenes (see Rules A-72 to A-75).
The radical name "phenyl" is retained.Examples to Rule A-11.4
11.5 - Names of bivalent radicals derived from saturated or unsaturated monocyclic hydrocarbons by removal of two atoms of hydrogen from the same carbon atom of the ring are obtained by replacing the endings "-ane", "-ene", "-yne", by "-ylidene", "-enylidene" and "-ynylidene", respectively. The carbon atom with the free valences is numbered as 1 except as stated in the rules for terpenes.
11.6 - Bivalent radicals derived from saturated or unsaturated monocyclic hydrocarbons by removing a hydrogen atom from each of two different :carbon atoms of the ring are named by replacing the endings "-ane", "-ene", "-diene", "-yne", etc., of the hydrocarbon name by "-ylene", "-enylene", "-dienylene", "-ynylene", etc., the positions of the double and triple bonds and of the points of attachment being indicated. Preference n lowest numbers is given to the carbon atoms having the free valences.Examples to Rule A-11.5
See Recommendations'93 R-2.3.1Examples to Rule A-11.6
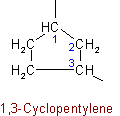
The following name is retained:


Published with permission of the IUPAC by Advanced Chemistry Development, Inc., www.acdlabs.com, +1(416)368-3435 tel, +1(416)368-5596 fax. For comments or suggestions please contact [email protected]