
ACD/Name (Chemist Version) offers a standardized set of features for quick and simple generation of IUPAC names, and structures from names. It is a streamlined version of our popular ACD/Name software.
View a full description and pricing on our web store.

13.2 - Since the name phenylene (o-, m- or p-) is retained for the radicalExamples to Rule A-13.1
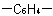 (exception to Rule A-11.6), bivalent radicals formed from
substituted benzene derivatives and having the free valences at ring atoms are named as substituted phenylene radicals. The carbon atoms having the free valences are numbered 1,2-, 1,3- or 1,4- as
appropriate.
(exception to Rule A-11.6), bivalent radicals formed from
substituted benzene derivatives and having the free valences at ring atoms are named as substituted phenylene radicals. The carbon atoms having the free valences are numbered 1,2-, 1,3- or 1,4- as
appropriate.
13.3-The following trivial names for radicals having a single free valence in the side chain are retained:
13.4 - Multivalent radicals of aromatic hydrocarbons with the free valences in the side chain are named in accordance with Rule A-4.Examples to Rule A-13.3
13.5 - The generic names of univalent and bivalent aromatic hydrocarbon radicals are "aryl" and "arylene", respectively.Examples to Rule A-13.4
See Recommendations'93 R-9.1, Table 19(b)


Published with permission of the IUPAC by Advanced Chemistry Development, Inc., www.acdlabs.com, +1(416)368-3435 tel, +1(416)368-5596 fax. For comments or suggestions please contact [email protected]