
ACD/Name (Chemist Version) offers a standardized set of features for quick and simple generation of IUPAC names, and structures from names. It is a streamlined version of our popular ACD/Name software.
View a full description and pricing on our web store.

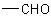 group attached to a carbon atom. Aldehydes corresponding to carboxylic acids with trivial names (see
R-9.1, Table 28(a)) are named by changing the "-ic acid" or "-ioc acid" ending of the acid name to "-aldehyde". Acyclic mono- and dialdehydes are named by adding the suffixes "-al"
or "c-dial" to the name of the acyclic hydrocarbon with the same number of carbon atoms, eliding the final "e" of the hydrocarbon name befor "a". Other aldehydes are named by adding the suffix
"-carbaldehyde" to the name of a parent hydride. When a group having priority for citation as a principal characteristic group is present, an aldehyde group is described by the prefix "formyl-". In
names of natural products, conversion of an implied CH3 group to an aldehyde is indicated by the prefix "oxo-".
group attached to a carbon atom. Aldehydes corresponding to carboxylic acids with trivial names (see
R-9.1, Table 28(a)) are named by changing the "-ic acid" or "-ioc acid" ending of the acid name to "-aldehyde". Acyclic mono- and dialdehydes are named by adding the suffixes "-al"
or "c-dial" to the name of the acyclic hydrocarbon with the same number of carbon atoms, eliding the final "e" of the hydrocarbon name befor "a". Other aldehydes are named by adding the suffix
"-carbaldehyde" to the name of a parent hydride. When a group having priority for citation as a principal characteristic group is present, an aldehyde group is described by the prefix "formyl-". In
names of natural products, conversion of an implied CH3 group to an aldehyde is indicated by the prefix "oxo-".
Chalcogen analogues of aldehydes are named using suffixes such as "-thial", "-selenal", "-carbothialdehyde", and "-carboselenaldehyde" and prefixes such as "thioformyl-" and "thioxo-"Examples to R-5.6.1
 . The use of the prefixes "thio-", "seleno-", and "telluro-" with trivial names of aldehydes such as acetaldehyde to indicate replacement of an aldehydic
oxygen atom is not included in these recommendations.
. The use of the prefixes "thio-", "seleno-", and "telluro-" with trivial names of aldehydes such as acetaldehyde to indicate replacement of an aldehydic
oxygen atom is not included in these recommendations.
Examples to R-5.6.1
4-Thioxocyclohexane-1-carboselenaldehyde
(see also R-5.6.2.2)



Published with permission of the IUPAC by Advanced Chemistry Development, Inc., www.acdlabs.com, +1(416)368-3435 tel, +1(416)368-5596 fax. For comments or suggestions please contact [email protected]