
ACD/Name (Chemist Version) offers a standardized set of features for quick and simple generation of IUPAC names, and structures from names. It is a streamlined version of our popular ACD/Name software.
View a full description and pricing on our web store.

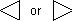

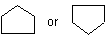

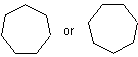
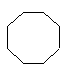
and the polycyclic system is oriented so that (a) the greatest number of rings are in a horizontal row and (b) a maximum number of rings are above and to the right of the horizontal row (upper right quadrant). If two or more orientations meet these requirements, the one is chosen which has as few rings as possible in the lower left quadrant.
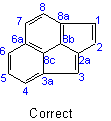
The system thus oriented is numbered in a clockwise direction commencing with the carbon atom not engaged in ring-fusion in the most counter-clockwise position of the uppermost ring, or if there is a choice, of the uppermost ring farthest to the right, and omitting atoms common to two or more rings.Example to Rule A-22.1
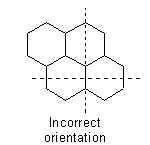
22.2 - Atoms common to two or more rings are designated by adding roman letters "a", "b", "c", etc., to the number of the position immediately preceding. Interior atoms follow the highest number, taking clockwise sequence wherever there is a choice.Examples to Rule A-22.1
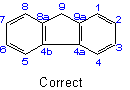
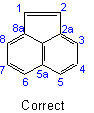
22.3 - When there is a choiceExamples to Rule A-22.2
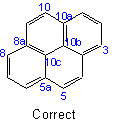
 , carbon atoms common to two or more rings follow the lowest possible numbers.
, carbon atoms common to two or more rings follow the lowest possible numbers.
22.4 - When there is a choice, the carbon atoms which carry an indicated hydrogen atom are numbered as low as possible.Examples to Rule A-22.3
Notes:
I 4, 4, 8, 9 is lower than 4, 5, 9, 9.
II 2, 5, 8 is lower than 3, 5, 8.
III 2, 3, 6, 8 is lower than 3, 4, 6, 8 or 2, 4, 7, 8.
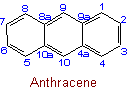
22.5 - The following are recommended exceptions to the above rules on numbering:Examples to Rule A-22.4
See Recommendations'93 R-2.4.1, R-9.1, Tables 20 and 20(a)Examples to Rule A-22.5



Published with permission of the IUPAC by Advanced Chemistry Development, Inc., www.acdlabs.com, +1(416)368-3435 tel, +1(416)368-5596 fax. For comments or suggestions please contact webmaster@acdlabs.com