
ACD/Name (Chemist Version) offers a standardized set of features for quick and simple generation of IUPAC names, and structures from names. It is a streamlined version of our popular ACD/Name software.
View a full description and pricing on our web store.

R-0.1.8.1 Nondetachable prefixes that modify the skeletal structure of a parent hydride are cited in alphabetical order immediately preceding
the name of the parent hydride. This type of prefix is found commonly in names of natural products (stereoparents) and is discussed more fully in Section F of the IUPAC Nomenclature of
Organic Chemistry ; however, they are occasionally encountered in trivial and semisystematic names, for example, homocubane.
; however, they are occasionally encountered in trivial and semisystematic names, for example, homocubane.
R-0.1.8.2 Nondetachable prefixes ("a" replacement terms commonly known as "a" prefixes) that indicate replacement of skeletal atoms of a parent hydride are cited in the
order of appearance in R-9.3, immediately preceding nondetachable skeleton-modifying prefixes (see R-0.1.8.1), if any, for example, 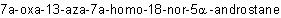 .
.
R-0.1.8.3 Detachable prefixes describing substituents are cited preceding nondetachable prefixes (see R-0.1.8.1 and R-0.1.8.2), if any, and are alphabetized as follows:
(a) Simple prefixes (i.e., those describing atoms and unsubstituted substituents) are arranged alphabetically; multiplying affixes, if necessary, are then inserted and do not alter the alphabetical order already established.
(b) The name of a prefix for a substituted substituent is considered to begin with the first letters of its complete name.Examples to R-0.1.8.3(a)
(c) When two or more prefixes consist of identical roman letters, priority for citation is given to that group which contains the lowest locant at the first point of difference.Example to R-0.1.8.3(b)
(Difluorobutyl as a complete substituent is alphabetized under "d".)
(d) o- (ortho), m- (meta), and p- (para) substituents, if otherwise identical, are arranged in that order (the same as their numerical equivalents, 2-, 3-, and 4-, respectively).Example to R-0.1.8.3(c)
R-0.1.8.4 Subtractive prefixes (such as anhydro-, dehydro-, demethyl-) have been used as "detachable" or "nondetachable" in previous editions of the IUPAC Nomenclature of Organic ChemistryExample to R-0.1.8.3(d)
 , but in these recommendations, they are presented as nondetachable.
, but in these recommendations, they are presented as nondetachable.
R-0.1.8.5 Additive prefixes. According to the previous edition of the IUPAC Nomenclature of Organic ChemistryExample to R-0.1.8.4
 (C-16.11), the additive prefixes "-dihydro-", "tetrahydro-", etc., could be either detachable and alphabetized among the set of substitutive prefixes, or nondetachable and
cites after the substitutive prefixes. In this guide these prefixes are presented as nondetachable
(C-16.11), the additive prefixes "-dihydro-", "tetrahydro-", etc., could be either detachable and alphabetized among the set of substitutive prefixes, or nondetachable and
cites after the substitutive prefixes. In this guide these prefixes are presented as nondetachable .
.
Example to R-0.1.8.5
4-Oxo-1,2,3,4-tetrahydronaphthalene-1-carboxylic acid



Published with permission of the IUPAC by Advanced Chemistry Development, Inc., www.acdlabs.com, +1(416)368-3435 tel, +1(416)368-5596 fax. For comments or suggestions please contact [email protected]