
ACD/Name (Chemist Version) offers a standardized set of features for quick and simple generation of IUPAC names, and structures from names. It is a streamlined version of our popular ACD/Name software.
View a full description and pricing on our web store.

 to a mononuclear hydride (see R-2.1) of the nitrogen, chalcogen, and
halogen families having a standard bonding number (see R-1.1) is named by adding the suffix "-onium" to a root for the element (see Table 17). Substituents are described in the
usual way.
to a mononuclear hydride (see R-2.1) of the nitrogen, chalcogen, and
halogen families having a standard bonding number (see R-1.1) is named by adding the suffix "-onium" to a root for the element (see Table 17). Substituents are described in the
usual way.
A cation derived formally by the addition of one or more hydrons to any position of a neutral parent hydride is described by replacing the final "e" of the parent hydride name, if any, by the suffix "-ium", or by adding the suffix "-ium", "-diium", etc., to the name of the parent hydride.Examples to R-5.8.2
Cations formally derived by the removal of one hydride ion,Examples to R-5.8.2
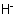 , from a parent hydride are named using the suffix "-ylium", in the same way as the suffix "-yl" (see
R-5.8.1.1). Di- and polycations formally derived by the removal of two or more hydride ions from a parent hydride are named by adding the suffixes "-bis(ylium) ", "-tris(ylium)", etc.,
to the name of the parent hydride. A cation that can be considered to be formally derived by the loss of an unpaired electron from the corresponding radical may also be named by adding the class name
"cation" as a separate word following the name of the radical.
, from a parent hydride are named using the suffix "-ylium", in the same way as the suffix "-yl" (see
R-5.8.1.1). Di- and polycations formally derived by the removal of two or more hydride ions from a parent hydride are named by adding the suffixes "-bis(ylium) ", "-tris(ylium)", etc.,
to the name of the parent hydride. A cation that can be considered to be formally derived by the loss of an unpaired electron from the corresponding radical may also be named by adding the class name
"cation" as a separate word following the name of the radical.
Cations formally derived by the loss of all the hydroxy groups as hydroxide ions from acid characteristic groups expressed by the suffix are named by replacing an "-ic acid" ending or suffix by "-ylium", or a "-carboxylic acid" suffix by "-carbonylium". Such cations may also be named by adding the class name "cation" as a separate word after the name of the acyl group.Examples to R-5.8.2
Prefix names for expressing monovalent monocationic parent cations as substituents with the free valence at the cationic site are formed by changing the "-ium" or "-onium" ending of the parent cation name to "-io" or "-onio", respectively.Examples to R-5.8.2
Prefixes for expressing substituent structural units containing cationic centres are derived systematically by adding the operational suffixes "-yl", "-ylidene", "-diyl", etc., together with appropriate locants, to the name of the parent cation.Examples to R-5.8.2
For replacement nomenclature, cationic replacement prefixes corresponding to "-onium" cations are formed, except for bismuth, by replacing the final "a" of the replacement prefix for the corresponding neutral heteroatom by "-onia", for example, "azonia-", "oxonia-", and "thionia-"; the cationic replacement prefix corresponding to bismuthonium is "bismuthonia-". Cationic replacement prefixes corresponding to "-ylium" cations are formed by replacing the final "e" of the name of the corresponding mononuclear parent hydride by "-ylia", for example, "azanylia" and "boranylia".Examples to R-5.8.2
Example to R-5.8.2



Published with permission of the IUPAC by Advanced Chemistry Development, Inc., www.acdlabs.com, +1(416)368-3435 tel, +1(416)368-5596 fax. For comments or suggestions please contact [email protected]