
ACD/Name (Chemist Version) offers a standardized set of features for quick and simple generation of IUPAC names, and structures from names. It is a streamlined version of our popular ACD/Name software.
View a full description and pricing on our web store.

 , are first named as "ortho-fused" or "ortho- and
peri-fused" systems. The other bridges are then indicated by prefixes derived from the name of the corresponding hydrocarbon by replacing the final "-ane", "-ene", etc., by "-ano", "-eno",
etc., and their positions are indicated by the points of attachment in the parent compound. If bridges of different types are present, they are cited in alphabetical order.
, are first named as "ortho-fused" or "ortho- and
peri-fused" systems. The other bridges are then indicated by prefixes derived from the name of the corresponding hydrocarbon by replacing the final "-ane", "-ene", etc., by "-ano", "-eno",
etc., and their positions are indicated by the points of attachment in the parent compound. If bridges of different types are present, they are cited in alphabetical order.
Examples of bridge names to Rule A-34.1
Butano Benzeno (o-, m-, p-) Ethano Etheno Methano Propano
34.2 - The parent "ortho-fused" or "ortho- and peri-fused" system is numbered as prescribed in Rule A-22. Where there is a choice, the position numbers of the bridgeheads should be as low as possible. The remaining bridges are then numbered in turn starting each time with the bridge atom next to the bridgehead possessing the highest number.Examples to Rule A-34.1
34.3 - When there is a choice of position numbers for the points of attachment for several individual bridges, the lowest numbers are assigned to the bridgeheads in the order of citation of the bridges and the bridge atoms are numbered according to the preceding rule.Example to Rule A-34.2
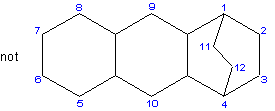
34.4 - When the bridge is formed from a bivalent cyclic hydrocarbon radical, low numbers are given to the carbon atoms constituting the shorter bridge and numbering proceeds around the ring.Example to Rule A-34.3
34.5 - Names for radicals derived from the bridged hydrocarbons considered in Rule A-34.1 are constructed in accordance with the principles set forth in Rule A-24. The abbreviated radical names naphthyl, anthryl, phenanthryl, naphthylene, etc., permitted as exceptions to Rules A-24.2 and A-24.4, are replaced in such cases by the regularly formed names naphthalenyl, anthracenyl, phenanthrenyl, naphthalenediyl, etc.Examples to Rule A-34.4
See Recommendations'93 R-2.4.1.2Examples to Rule A-34.5


Published with permission of the IUPAC by Advanced Chemistry Development, Inc., www.acdlabs.com, +1(416)368-3435 tel, +1(416)368-5596 fax. For comments or suggestions please contact [email protected]