
ACD/Name (Chemist Version) offers a standardized set of features for quick and simple generation of IUPAC names, and structures from names. It is a streamlined version of our popular ACD/Name software.
View a full description and pricing on our web store.

472.1 - When the corresponding hydroxy acid from which water may be considered to have been eliminated has a trivial name, the lactone is designated by substituting "-olactone" for "-ic acid" in the name of the hydroxy acid. Any necessary locants are added, that for a carbonyl group being as low as possible. If the locants for both the hydroxyl and the carbonyl group require to be cited, that for the carbonyl group is placed first.
472.2 - Lactones formed from aliphatic acids are named by adding "-olide" to the name of the (non-hydroxylated) hydrocarbon with the same number of carbon atoms, with elision of terminal "e", if present, before a vowel. Locants are added to define the position of ring closure.Examples to Rule C-472.1
Note: "-olide" signified the change ofExamples to Rule C-472.2
 into
into 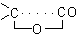
For naming certain lactones of hydroxy acids in the steroid series see the Rules for Nomenclature of Steroids, Pure and Applied Chemistry, 1972, Vol.31, Nos.1-2 (Rules 2S-3 and 2S-4).
472.3 - Structures in which one or more (but not all) rings of an aggregate are lactone rings are named by placing "-carbolactone" (denoting the -O-CO- bridge) after the names of the structures which remain when each -O-CO- is replaced by two hydrogen atoms. Multiplying affixes "bis-", "tris- '', etc., and/or locants are used as necessary (see Rule C-472.1). The locant for -CO- is cited before that for the ester -O-.
Note: "carbolactone" signified the change ofExample to Rule C-472.3
 into
into  , an additional carbon atom being incorporated.
, an additional carbon atom being incorporated.472.4 - The following common names, derived from trivial names of non-hydroxylated acids, are permitted and may be used in naming their substitution products, but the principle of their formation should not be extended to other lactones:
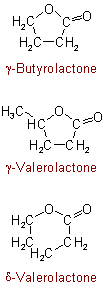
See Recommendations'93 R-5.7.5
Next:
Lactones, Lactides, Lactams, and Lactims Rule C-473, Rule C-474, Rule C-475
Acyl Halides Rule C-481
Acid Anhydrides Rule C-491




Published with permission of the IUPAC by Advanced Chemistry Development, Inc., www.acdlabs.com, +1(416)368-3435 tel, +1(416)368-5596 fax. For comments or suggestions please contact [email protected]