
ACD/Name (Chemist Version) offers a standardized set of features for quick and simple generation of IUPAC names, and structures from names. It is a streamlined version of our popular ACD/Name software.
View a full description and pricing on our web store.


The suffix '-ol' with its multiplicative prefix 'di-' and locants '1' and '11' indicates the presence of two hydroxy (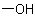 ) groups at positions 1 and 11 of undecane. The infixes '-ene' and '-yne', with
their multiplicative prefix 'di-' and locants '2' and '4', and '7' and '9', describe double bonds at positions 2 and 4 and triple bonds at positions 7 and 9, respectively.
) groups at positions 1 and 11 of undecane. The infixes '-ene' and '-yne', with
their multiplicative prefix 'di-' and locants '2' and '4', and '7' and '9', describe double bonds at positions 2 and 4 and triple bonds at positions 7 and 9, respectively.
The resulting parent structure with its numbering is:
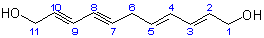
(b) The complex prefix '(4-hydroxyhex-1-en-1-yl-)' with its locant '6' describes a substituent at position 6 of the parent structure. The parent substituent is a six-carbon chain corresponding to the parent
hydride name 'hexane'. The prefix 'hydroxy-' with its locant '4' denotes a hydroxy 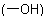 group at position 4; and the infix '-en-' with its locant '1' and the suffix '-yl-' with its locant '1'
describe, respectively, a double bond at position 1 and a free valence at position 1. Hence, the structure and numbering of the complex substituent is:
group at position 4; and the infix '-en-' with its locant '1' and the suffix '-yl-' with its locant '1'
describe, respectively, a double bond at position 1 and a free valence at position 1. Hence, the structure and numbering of the complex substituent is:
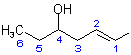
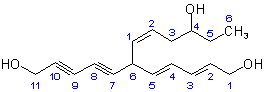
(c) The structure of the complete compound is therefore:
 -methylquinoline-2-pentanoic acid
-methylquinoline-2-pentanoic acid



Published with permission of the IUPAC by Advanced Chemistry Development, Inc., www.acdlabs.com, +1(416)368-3435 tel, +1(416)368-5596 fax. For comments or suggestions please contact [email protected]