

ACD/Name (Chemist Version) offers a standardized set of features for quick and simple generation of IUPAC names, and structures from names. It is a streamlined version of our popular ACD/Name software.
View a full description and pricing on our web store.
In Section D, this system is broadened by extending Table B-I of Rule B-1 to include most of the elements. The corresponding "a" terms are given in Table D-I. This seniority order of elements is based upon the element sequence given in Table IV of "Nomenclature of Inorganic Chemistry, 1970", and supersedes Rule B-1.4.
1.61 - The affixes listed in Table D-I in decreasing order of priority are used according to the principles stated in Subsection C-0.6 for acyclic structures and in Rule B-4 for heterocyclic systems. These prefixes are used alone when the heteroatoms considered in the structure have the normal bonding number. The accepted normal bonding numbers are the following:
 |
|
| Elements | Normal bonding number |
 |
|
| F, Cl, Br, I, At | 1 |
| O, S, Se, Te, Po | 2 |
| N, P, As, Sb, Bi | 3 |
| C, Si, Ge, Sn, Pb | 4 |
| B, Al, Ga, In, Tl | 3 |
| Be, Mg, Zn, Cd, Hg | 2 |
1.62 - When the bonding number of a neutral heteroatom is other than that given in Rule D-1.61 or if the heteroatom is not listed under that rule, the bonding number is expressed by an Arabic number cited as a superscript to the Greek letterExamples to Rule D-1.61
1-Methyl-1-aluminacyclohexane
Methyl 3-phospha-5-silahexanoate
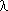 which follows the locant for the heteroatom. If no locant for the heteroatom normally appears in the name
for the parent compound, the l symbol and an appropriate locant are prefixed to that part of the name which designates the heteroatom.
which follows the locant for the heteroatom. If no locant for the heteroatom normally appears in the name
for the parent compound, the l symbol and an appropriate locant are prefixed to that part of the name which designates the heteroatom.
Note: - In some cases, bonding numbers higher than those listed in Rule D-1.61 may be indicated by hydro prefixes according to Rule D-1.53.
1.63 - Ionic heteroatoms are expressed, whenever possible, by the usual terms such as "-onium", "-ium", "-ide", "-onia", "-ata", with their usual meaning (see Subsections C-0.8 and D-7.63) associated with normal bonding numbers.Examples to Rule D-1.62
Examples to Rule D-1.63
Tetramethylphosphonium bromide
Pyridinium perchlorate
Sodium 4a-borataanthracene


Published with permission of the IUPAC by Advanced Chemistry Development, Inc., www.acdlabs.com, +1(416)368-3435 tel, +1(416)368-5596 fax. For comments or suggestions please contact [email protected]