Bring the power of IUPAC naming to your desktop!
ACD/Name (Chemist Version) offers a standardized set of features for quick and simple generation of IUPAC names, and structures from names. It is a streamlined version of our popular ACD/Name software.
View a full description and pricing on our web store.
Groups Containing One Nitrogen Atom
Amines Rule C-811 General 
811.1 - Bases in which nitrogen forms part of a ring are named as heterocyclic compounds in accordance with Section B of
these Rules. The termination "-ine", "-ole", or "-ete" of such names is not considered to be a suffix in the sense of Rule C-10.3, and so may be followed by
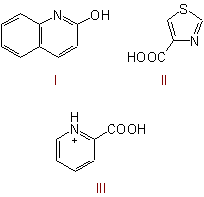
endings to denote a substituent of the heterocycle, as, for example, in 2-quinolinol (I) or 4-thiazolecarboxylic acid (II). However, the endings denoting quaternary atoms (see Rule C-816)
are considered as suffixes, and substituents must then be named by prefixes, as, for example, in 2-carboxypyridinium chloride (III). In the same way as "-ium" the ending "-amine" (see below) is
considered as a suffix.
811.2 - The generic name "amine'' is applied to compounds NH2R, 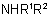 , and
, and 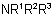 , which are called primary, secondary, and
tertiary amines, respectively. In a wider sense, compounds containing nitrogen in a ring and owing their basicity to this atom may also be referred to as "amines".
, which are called primary, secondary, and
tertiary amines, respectively. In a wider sense, compounds containing nitrogen in a ring and owing their basicity to this atom may also be referred to as "amines".
811.3 - An -NH2 group, when not the principal group, is named by the prefix "amino-".
Examples to Rule C-811.3
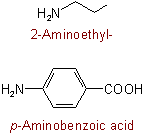
Note: In Beilstein's Handbuch der organischen Chemie use of the prefix "amino-" is permitted as an alternative, when -NH2 is the principal group, but this method is not recommended
here; it leads to names such as 1-aminobutane, 2-aminopentane, and 1-aminoanthracene.
811.4 - Radicals RNH-, R2N-, and 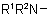 are named as substituted amino groups by changing "ine'' of the amine to "ino".
are named as substituted amino groups by changing "ine'' of the amine to "ino".
Examples to Rule C-811.4
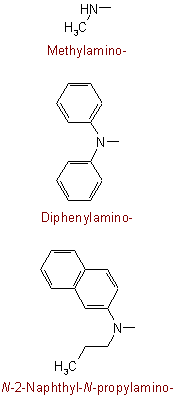
The following are examples of trivial names which are retained:
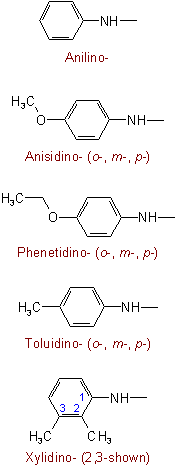
See Recommendations'93 R-5.4
- Next:
-
Amines C-812, C-813, C-814, C-815,
C-816
Amides and Imides C-821, C-822, C-823,
C-824, C-825, C-826, C-827
Nitriles, Isocyanides, and Their Derivatives C-831, C-832, C-833,
C-834
Hydroxylamines and Related Compounds C-841, C-842, C-843
Nitroso and Nitro Compounds C-851, C-852
Amine Radical Ions C-861


This HTML reproduction of Sections A, B and C of IUPAC "Blue Book" is as close as possible to the
published version [see Nomenclature of Organic Chemistry, Sections A, B, C, D, E, F, and H, Pergamon Press, Oxford, 1979.
Copyright 1979 IUPAC.] If you need to cite these rules please quote this reference as their source.
Published with permission of the IUPAC by Advanced Chemistry Development, Inc., www.acdlabs.com, +1(416)368-3435 tel, +1(416)368-5596 fax. For comments or suggestions please contact [email protected]



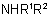 , and
, and 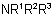 , which are called primary, secondary, and
tertiary amines, respectively. In a wider sense, compounds containing nitrogen in a ring and owing their basicity to this atom may also be referred to as "amines".
, which are called primary, secondary, and
tertiary amines, respectively. In a wider sense, compounds containing nitrogen in a ring and owing their basicity to this atom may also be referred to as "amines".
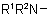 are named as substituted amino groups by changing "ine'' of the amine to "ino".
are named as substituted amino groups by changing "ine'' of the amine to "ino".

