
ACD/Name (Chemist Version) offers a standardized set of features for quick and simple generation of IUPAC names, and structures from names. It is a streamlined version of our popular ACD/Name software.
View a full description and pricing on our web store.

832.1 - Compounds RCN, in which 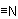 replaces H3 at the end of the main chain of an acyclic hydrocarbon are denoted by adding "-nitrile" or
"-dinitrile" to the name of this hydrocarbon.
replaces H3 at the end of the main chain of an acyclic hydrocarbon are denoted by adding "-nitrile" or
"-dinitrile" to the name of this hydrocarbon.
Note: "Nitrile'', here and in Rule C-832.3, denotes the triply bound nitrogen atom,Examples to Rule C-832.1
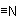 , and not the carbon atom attached to it. Numbering begins with that carbon
atom.
, and not the carbon atom attached to it. Numbering begins with that carbon
atom.
832.2 - Compounds RCN, when considered as derived from acids R-COOH whose systematic names end in "-carboxylic acid", are named by changing this ending to "-carbonitrile".
Note: "Carbonitrile'' denotes the group 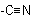 , including the carbon atom contained therein. That carbon atom is excluded from the numbering of a chain to which that group is
attached.
, including the carbon atom contained therein. That carbon atom is excluded from the numbering of a chain to which that group is
attached.
832.3 - Names of compounds RCN, when considered as derived from acids R-COOH having trivial names, are formed by changing the syllables "-oic acid" to "-onitrile", or, if the name of the acid does not end in "-oic acid", then by changing "-ic acid" to "-onitrile".Examples to Rule C-832.2
832.4 - By the radicofunctional procedure, compounds RCN are named by stating the name of the radical R, followed by the name "cyanide" for the group -CN.Examples to Rule C-832.3
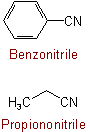
(but see Rules C-832.1 and C-832.4)
832.5 - When the compound contains also a group that has priority over -CN for citation as principal group, the -CN group is named by the prefix "cyano-".Examples to Rule C-832.4
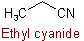
(but see Rules C-832.1 and C-832.3)
See Recommendations'93 R-5.7.9Examples to Rule C-832.5


Published with permission of the IUPAC by Advanced Chemistry Development, Inc., www.acdlabs.com, +1(416)368-3435 tel, +1(416)368-5596 fax. For comments or suggestions please contact [email protected]