Bring the power of IUPAC naming to your desktop!
ACD/Name (Chemist Version) offers a standardized set of features for quick and simple generation of IUPAC names, and structures from names. It is a streamlined version of our popular ACD/Name software.
View a full description and pricing on our web store.
Carboxylic Acids and their Derivatives
Amino Acids Rule C-421 

421.1 - Table VIII gives examples of trivial names for -amino
carboxylic acids. The name of the radical obtained by removal of hydroxyl from the carboxyl group is derived from the trivial name of the amino acid by replacing the ending "-ine" with "-yl''.
421.2 - The following names are also recommended, the radical names being exceptions to the preceding rule:
| Cysteine |
Cysteinyl |
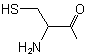 |
| Homocysteine |
Homocystenyl |
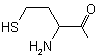 |
| Tryptophan |
Tryptophyl |
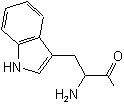 |
421.3 - (a) 2-Aminobutanedioic acid is named aspartic acid. (b) 2-Aminopentanedioic acid is named glutamic acid. (c) The
derived univalent acyl radicals are named aspartyl and glutamyl, while the corresponding bivalent acyl radicals are named aspartoyl and glutamoyl (compare Rule C-403.1 and
C-405.2). (d) Monoamides of these acids in which the carboxyl group numbered I is intact are respectively named asparagine and glutamine, and univalent acyl radicals corresponding
to these monoacids are named asparaginyl and glutaminyl (see Table IX).
421.4 - The following trivial names are also retained:
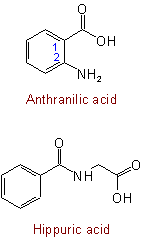
See Recommendations'93 R-5.7.1
- Next:
-
Amic acids Rule C-431
Peroxy Acids Rule C-441
Imidic, Hydrazonic, and Hydroxamic Acids Rule C-451
Salts and Esters Rule C-461, Rule C-462, Rule C-463,
Rule C-464
Lactones, Lactides, Lactams, and Lactims Rule C-471, Rule C-472, Rule C-473,
Rule C-474, Rule C-475
Acyl Halides Rule C-481
Acid Anhydrides Rule C-491


This HTML reproduction of Sections A, B and C of IUPAC "Blue Book" is as close as possible to the
published version [see Nomenclature of Organic Chemistry, Sections A, B, C, D, E, F, and H, Pergamon Press, Oxford, 1979.
Copyright 1979 IUPAC.] If you need to cite these rules please quote this reference as their source.
Published with permission of the IUPAC by Advanced Chemistry Development, Inc., www.acdlabs.com, +1(416)368-3435 tel, +1(416)368-5596 fax. For comments or suggestions please contact webmaster@acdlabs.com









