Bring the power of IUPAC naming to your desktop!
ACD/Name (Chemist Version) offers a standardized set of features for quick and simple generation of IUPAC names, and structures from names. It is a streamlined version of our popular ACD/Name software.
View a full description and pricing on our web store.
Carboxylic Acids and their Derivatives
Simple Carboxylic Acids Rule C-403 
403.1 - When the acid is named according to Rule C-401.1, the name of a univalent or bivalent acyl radical formed by removal of
hydroxyl from all the carboxyl groups is derived from the name of the corresponding acid by changing the ending "-oic'' to "-oyl''.
Examples to Rule C-403.1

403.2 - If an acid name is formed with the ending "-carboxylic", the name of the radical obtained by removal of hydroxyl from all carboxyl groups is formed by changing the
ending "-carboxylic" to "-carbonyl".
Examples to Rule C-403.2
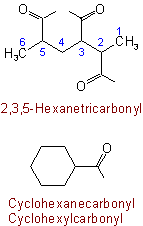
Note: In radicofunctional nomenclature, names of the type cyclohexanecarbonyl are used, as in:

When, however, the acyl group is a substituent, the compound radical prefix form of type cyclohexylcarbonyl is used, as in:
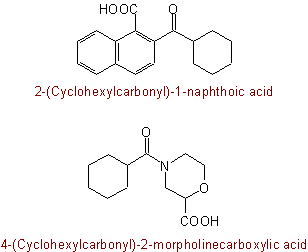
These principles apply to all acyl radicals whose names end in "carbonyl'', "sulfonyl'', "sulfinyl'', etc. (cf. Rules C-543.3, C-631.1, C-641.7,
C-641.8, and C-641.9).
See Recommendations'93 R-5.7.1
- Next:
-
Simple Carboxylic Acids Rule C-404, Rule C-405
Hydroxy, Alkoxy and Oxo Acids Rule C-411, Rule C-415, Rule C-416
Amino Acids Rule C-421
Amic acids Rule C-431
Peroxy Acids Rule C-441
Imidic, Hydrazonic, and Hydroxamic Acids Rule C-451
Salts and Esters Rule C-461, Rule C-462, Rule C-463,
Rule C-464
Lactones, Lactides, Lactams, and Lactims Rule C-471, Rule C-472, Rule C-473,
Rule C-474, Rule C-475
Acyl Halides Rule C-481
Acid Anhydrides Rule C-491



This HTML reproduction of Sections A, B and C of IUPAC "Blue Book" is as close as possible to the
published version [see Nomenclature of Organic Chemistry, Sections A, B, C, D, E, F, and H, Pergamon Press, Oxford, 1979.
Copyright 1979 IUPAC.] If you need to cite these rules please quote this reference as their source.
Published with permission of the IUPAC by Advanced Chemistry Development, Inc., www.acdlabs.com, +1(416)368-3435 tel, +1(416)368-5596 fax. For comments or suggestions please contact webmaster@acdlabs.com





