
ACD/Name (Chemist Version) offers a standardized set of features for quick and simple generation of IUPAC names, and structures from names. It is a streamlined version of our popular ACD/Name software.
View a full description and pricing on our web store.


The terminal "e" in names of parent compounds is elided when followed by a suffix beginning with "a", "I", or "o":
| Examples: | Cf. Rule |
| 4-Penten(e)-1-ol | C-10.3 |
| 2-Hexan(e)amine | C-11.42 |
| Anilin(e)ium | C-82.2 |
| Butan(e)ide | C-84.3 |
| 1-Chrysen(e)ol | C-202.1 |
| Hexan(e)al | C-302.1 |
| 2-Butan(e)one | C-312.1 |
| Heptan(e)oic acid | C-401.1 |
| 5-Pentan(e)olide | C-472.2 |
| 1-Hexan(e)imine | C-815.3 |
| Hexan(e)amide | C-822.1 |

The terminal "a" in the names of multiplying affixes is elided when followed by a suffix or ending (not a separate word) beginning with "a" or "o":
| Examples: | Cf. Rule |
| [1,1'-Binaphthalene]-3,3',4,4'-tetramine | C-71.3 |
| 1,3,6,8(2H,7H)-Pyrenetetrone | C-315.1 |
| Benzenehexol |

There is no elision in the following cases:
| (1) in conjunctive nomenclature, | Cf. Rule |
| e.g., Quinolineacetic acid | C-51.2 |
 , ,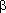 -Dimethyl-2-naphthaleneethanol -Dimethyl-2-naphthaleneethanol |
C-52.1 |
| (2) in replacement nomenclature, | |
| e.g., Tetraoxatridecane | C-61.1 |
| (3) in multiplying parent compounds, | |
| e.g., Ethylenediaminetetraacetic acid | C-815.1 |
| (4) with multiplying prefixes, | |
| e.g., 1,4,6,9-Tetraoxo-2-pyrenecarboxylic acid |
(6) For elision of vowels in forming names of fused polycyclic compounds see Rule A-21.4 and in Hantzsch-Widman names see Rule B-1.1.
See Recommendations'93 R-0.1.7



Published with permission of the IUPAC by Advanced Chemistry Development, Inc., www.acdlabs.com, +1(416)368-3435 tel, +1(416)368-5596 fax. For comments or suggestions please contact webmaster@acdlabs.com