January 26, 2023
by Arvin Moser, Team Manager, Application Scientists, ACD/Labs
1H NMR is a powerful technique for the elucidation and verification of the chemical structure of compounds. The peak position, or chemical shift, in 1H NMR spectra depends on the electron density around the proton. Higher electron density shifts the peak upfield to a lower ppm value, while lower electron density shifts the peak downfield to a higher ppm. Often in an 1H NMR spectrum, protons of a given functional group appear within an expected chemical shift range. However, this is not the case for protons that are covalently bonded to oxygen (-OH) or nitrogen (-NH). These protons undergo a fast exchange with other protons or deuterium in solution. This chemical process continuously replaces the protons in the solute molecules and affects their detection, chemical shift, and peak shape relative to other protons in the compound.
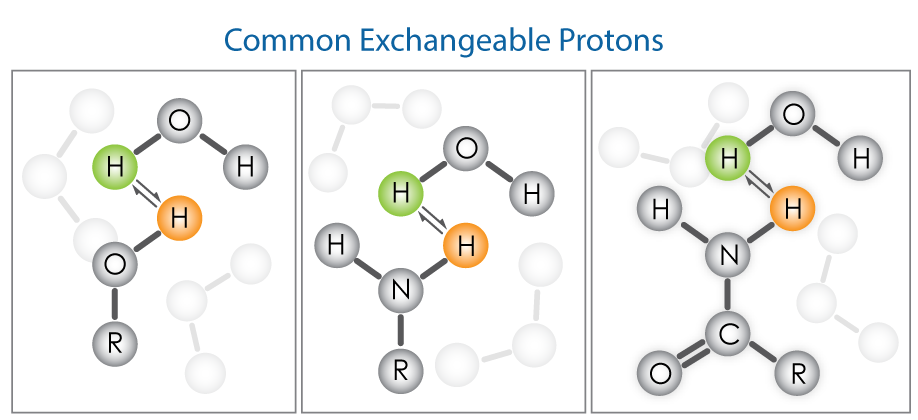
Exchangeable proton peaks can share the following characteristics:
- Wide range of chemical shifts: Many factors such as solvent, hydrogen bond strength, pH, temperature, and concentration can strongly impact the chemical shift of exchangeable protons. For example, the formation of H-bonds leads to a significant shift to higher frequency. Therefore, resonances of exchangeable protons may appear at different chemical shifts. In other words, there is no ‘absolute correct position’ for these signals and experimental conditions determine where they appear. This can cause some confusion during peak assignment. Typical 1H chemical shift ranges for OH and NH protons are shown in the following table:

- Broad peaks: In protic solvents, the exchangeable protons readily exchange at room temperature and result in a broader peak which is an average of all the signals. In this case, reducing the temperature can reduce the exchange rate, and relatively sharper peaks appear.
- Splitting may or may not be observed on the peaks: Since these protons rapidly exchange between molecules, the adjacent protons may not always ‘feel their presence’, and vise versa. Thus, the peaks appear without the expected coupling.
Solvent Effects on Exchangeable Proton Peaks
The broad peaks of exchangeable protons can overlap with other peaks in the sample and obscure the information. Fortunately, experimental conditions (solvents, concentrations, temperature, etc.) can be used to control this. For example, the type of deuterated solvents can determine if exchangeable protons are observed in the 1H NMR spectrum. The presence of an exchangeable proton peak depends on the exchange rate with the protons or deuteria from the bulk solvent.
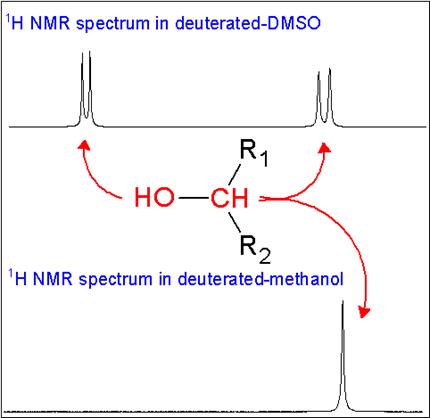
Here we compare the 1H NMR spectra of a secondary alcohol, HOCHR2, dissolved in DMSO-d6 and deuterated methanol. Proton exchange rates are reduced in DMSO-d6 and there are no exchangeable deuterium atoms in the solvent. Therefore the 1H NMR spectrum shows the expected doublet for the hydroxyl proton and another doublet for methine proton. The spectrum acquired in deuterated methanol shows only a singlet for the CH proton, due to the relatively high proton exchange rate. The OH proton peak is not observed because of its exchange with deuterium atoms from the solvent.
Exchangeable Protons Used in Structural Studies
Exchangeable protons provide valuable insights into macromolecular structure and dynamics. Hydrogen/deuterium (H/D) exchange combined with 2D NMR has been widely applied to investigate the structure, stability, and dynamics of proteins. For example, the proton in the amide groups of the backbone of amino acids can be deliberately replaced (labeled) with deuterium. The labeled sites can then be used to identify the solvent accessible residues and provide information on non-native states of proteins, such as equilibrium and kinetic folding intermediates.
Relaxation techniques such as Nuclear Overhauer Effect Spectroscopy (NOESY) and Rotating frame Overhauser Effect Spectroscopy (ROESY) can be used to study proton exchange rates in proteins. For protons undergoing chemical exchange, corresponding cross peaks are observed in these spectra. These experiments measure the rate at which the exchangeable protons relax back to the equilibrium state. This information can reveal the proximity of the protons to each other and assist in revealing the 2D or 3D structure.
Recognizing Exchangeable Protons in NMR Spectra
If you intend to retain the exchangeable proton resonances or work out the spectra containing such protons, keep in mind that a key to interpreting a 1H NMR spectrum is to correctly distinguish between CHn protons (where n = 1, 2, or 3) and exchangeable protons. Acquiring a 2D 1H-13C correlation spectra, such as HMQC, HSQC or HETCOR, can help in assigning the peaks.
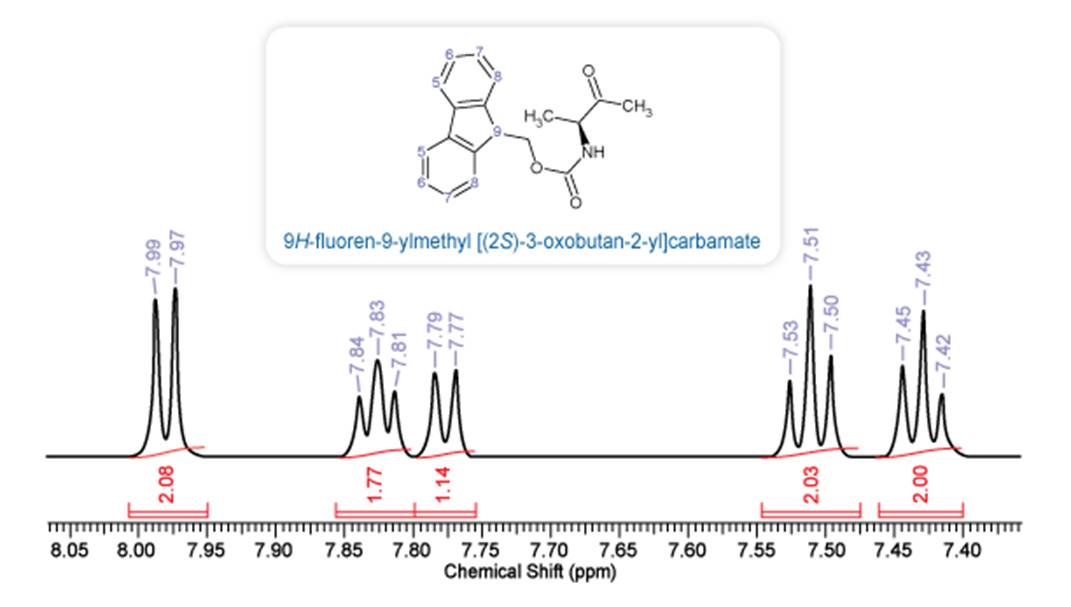
To demonstrate this using an example, we look at the partial 1H NMR spectrum (7.40–8.05 ppm) of a carbamate compound with the structure shown below. The aromatic region shows 5 multiplets with a 2:2:1:2:2 ratio of relative intensities. The multiplets with integrals of 2 correspond to the aromatic protons from the fused ring system. The doublet (d) at 7.78 ppm may either correspond to a CH or NH proton. The chemical shift and coupling pattern (d) provide clues to the assignment, however, in this case the coupling constant for both doublets is (~10 Hz), making it impossible to distinguish the peaks using 1H NMR alone. An additional HSQC spectrum can be recorded to either rule out or confirm that the observed peak corresponds to an -OH or -NH proton.
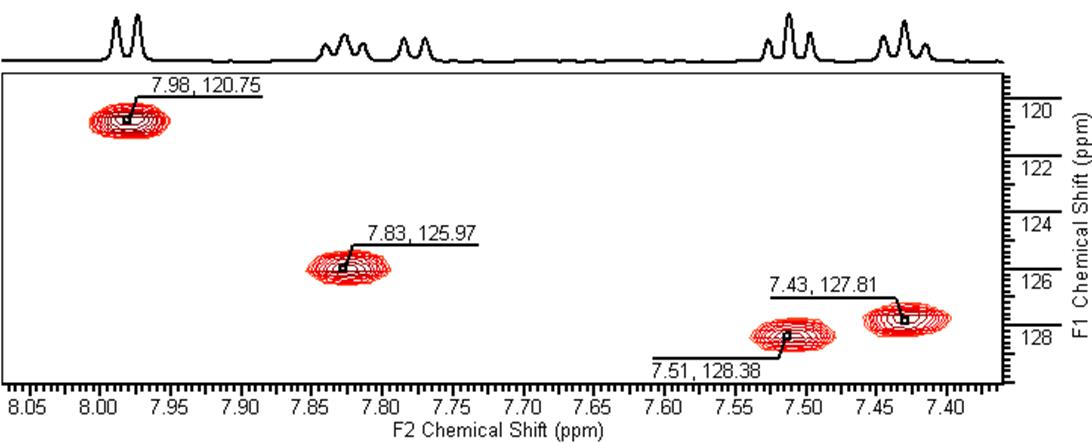
The HSQC spectrum correlates 1H-13C groups separated by a single bond. The absence of a cross peak for the doublet at 7.78 ppm rules out the presence of a CH group and suggests that the doublet corresponds to an ‐OH or -NH proton.
Tip: There may be additional value in using a database or library to compare the data against the 1H NMR spectra of previously identified compounds with similar structures. For a list of available NMR databases, please visit here.
Structure Elucidation of Compounds with Exchangeable Protons
In order to successfully elucidate a structure, you need to extract the correct information from the 1H NMR spectrum. This information includes the total number of protons and the presence of exchangeable protons, where possible. When the exchangeable proton peaks are eliminated from the spectra of a given compound, a discrepancy between the total proton count from the spectra and the molecular formula arises. Based on the molecular formula and spectra for other nuclei, these non-exchangeable protons can be assigned to the atoms to which they are bound, leaving the elucidator with extra “floating” protons.
This can be illustrated when solving the correct structure with MF=C10H15N, as shown below. Initially the carbons were assigned as C, CH, CH2 or CH3, leaving three unassigned protons. Counting the expected number of exchangeable protons can serve as a warning flag that something is amiss. Assuming there is no ammonia (NH3) impurity in the compound, the carbon assignments were done incorrectly and should be reinvestigated by the user.
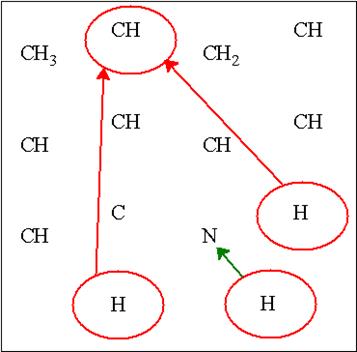
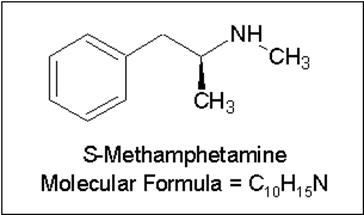
The correct structure of methamphetamine reveals that a methyl (CH3) was incorrectly set to a methine (CH) group.
Exchangeable protons may pose challenges in NMR data interpretation, but by selecting the proper experimental conditions their signals can be readily removed from the spectra. In many cases, only a small amount of D2O can eliminate the existing contributions and simplify spectral interpretation. Although you may not even want to eliminate these signals, if you can get them to work in your favor. With the right experimental conditions, exchangeable protons can and have been long used to get complementary information on the structure or dynamics of the molecules.


I want to gain more knowledge on Exchangeable protons in structural elucidation.
Hello Nagaraju,
You can learn more about exchangeable protons and the corresponding NMR data in more of our blog posts on the topic:
https://www.acdlabs.com/blog/how-do-i-know-w/
https://www.acdlabs.com/blog/interpreting-1/
https://www.acdlabs.com/blog/identifying-exchangeables-protons-on-a-1h-nmr-spectrum/