The Challenge of Heterogeneous Analytical Data
Pfizer is a global biopharmaceutical company with a portfolio of small molecule and biological R&D projects spanning diverse diseases. Analytical data plays a critical role in decision-making across drug discovery, drug product development, and manufacturing.
Pfizer scientists generate vast volumes of analytical data across multiple sites, techniques, and instrument vendor platforms, that are used in various informatics systems including LIMS, ELN, and mobile devices. Data heterogeneity makes analytical data difficult to find, reuse, and leverage.
Reliance on Manual Data Transfer Leads to Inefficiency & Risk
Prior to 2019, the analytical data landscape in Pfizer’s PharmSci small molecule group was typically heterogeneous. Chromatography, NMR, mass spectrometry, and solid-state data were stored across instruments, shared drives, desktops, and emails. Scientists relied heavily on personal expertise and institutional memory to locate historical results; often repeating experiments because prior data could not be found or reused.

As one Pfizer scientist described it:
“We had data scattered everywhere—on instruments, on servers, on laptops, even in people’s minds. Finding the right information meant finding the right person.”
– Vijay Bulusu (Head, Data & Digital Innovation, PharmSci, WRD, Pfizer)
These limitations created inefficiency with scientists spending too much time looking for data. It also increased operational risk and restricted Pfizer’s ability to use their data for meaningful visualization, modeling, and data science.
A Digital Evolution for Analytical Data Management at Pfizer
Pfizer’s initial objective was deliberately focused—to centralize chromatography data from their Empower CDS in a vendor-neutral environment and make it searchable across sites and projects. With the help of ACD/Labs technology, Pfizer established a Scientific Data Cloud that automated data ingestion, enriched records with chemical context and metadata, and standardized analytical knowledge for enterprise-wide access.
“What began as a simple goal—to centralize Empower data—soon revealed much bigger potential.”
– Bo Du, System Architect, Worldwide Research & Development, Pfizer
Pfizer’s journey towards digitalized analytical data management began by harmonizing data on the Spectrus Platform. Pfizer empowered their scientists with simpler processes and digitalized FAIR data. This resulted in reduced reliance on manual data movement and created a foundation that could scale to all their major analytical techniques and workflows.
Accelerating Structure Elucidation with Effective Management of NMR and MS Data
Pfizer extended the same model to NMR and mass spectrometry workflows, replacing email-based exchanges and fragmented vendor tools with a centralized, searchable environment. High-quality datasets can be promoted to curated “golden data” collections for training and reference.
“Mass spec and NMR results used to live in emails and network folders. Now they’re structured, searchable, and available globally. Spectrus provides easy, fast, automated, and central access to the data and connects different databases within Pfizer.”
– Bo Du, Pfizer
Data managed on the Spectrus Platform provides subject matter experts, project teams, and the Pfizer scientist community with:
- Global access to organized data
- Data that is searchable by Pfizer number (PF#), structure, substructure, and spectral match
- Automated workflows that ease project tracking and increase efficiency
Integrating Solid-State and Drug Product Analytics
Powder X-ray diffraction, DSC, TGA, and microscopy data are now integrated into unified dashboards, supporting formulation scientists and drug product teams with consistent, contextual dashboards of solid-state analyses.
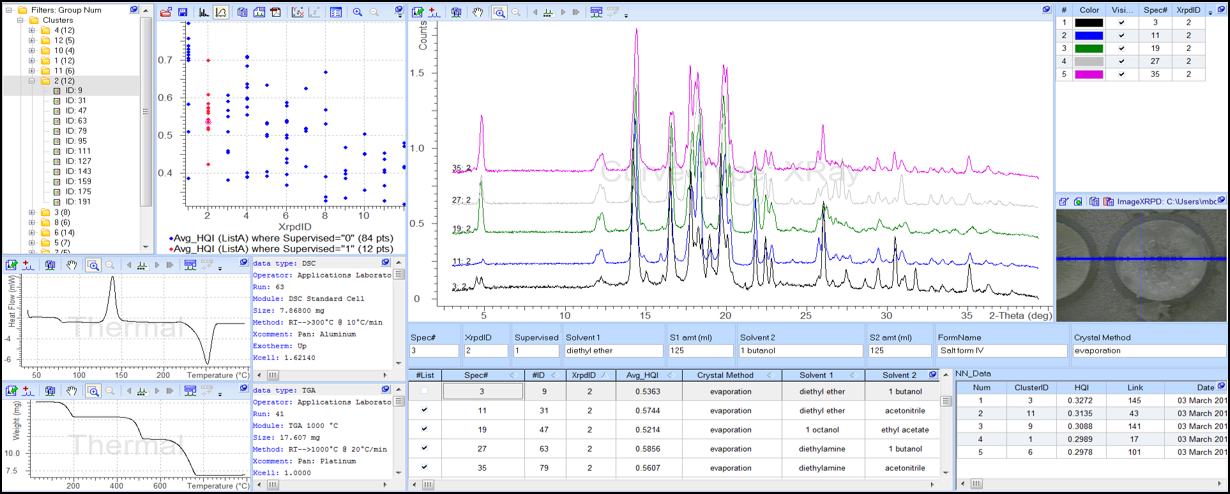
The drug product development dashboard at Pfizer combines DSC, powder x-ray data, microscopy images and other solid-state analyses.
“Powder X-ray, DSC, TGA, microscopy—these used to sit in different places. Now they’re part of the same analytical record.”
– Bo Du, Pfizer
Data can be accessed by multiple people simultaneously and is automatically associated with electronic notebooks records.
Knowledge-Centric Decision Support in CMC
Using Luminata software, Pfizer connects analytical evidence to process maps, impurity tracking, degradation studies, and stability data—creating a persistent knowledge base that supports complex process development decisions. Analytical data collected by the Structure Elucidation Group is connected with each synthetic step in the process—products, by-products, and impurities.
“Building the [Luminata] record takes time, but once it’s built, it transforms how we manage impurities, track batch genealogy, manage stability studies, and much more.”
– Bo Du, Pfizer
High-Throughput Experimentation
Pfizer is also piloting integrated high-throughput experimentation workflows, enabling standardized calculations, faster analysis, and consistent reporting across large experiment sets.
“In the past, everyone calculated results differently. Now the software gives us consistency—and saves days of manual work.”
– Bo Du, Pfizer
Improving Data Access & Reuse
By consolidating analytical data into a harmonized scientific data cloud, Pfizer has significantly improved:
- Global access to analytical results
- Reuse of historical data across projects and sites
- Efficiency in structure elucidation and impurity analysis
- Data consistency for modeling, AI, and machine learning
- Collaboration between internal teams and external partners
What began as a chromatography initiative has evolved into a scalable platform that supports multiple analytical domains and downstream digital initiatives.
“The integrated workflow accelerates our data reuse, our analysis, and our AI goals for the future.”
– Bo Du, Pfizer
Scaling Digitalized Data Management
Pfizer continues to expand its analytical data strategy, with plans to centralize data from additional CDS sources, extend solid-state dashboards, and introduce Spectrus JS thin-client applications to simplify software deployment and adoption.
“Our goal is to have all analytical data in one place—with simple access for everyone.”
– Bo Du, Pfizer
By investing in a data foundation, Pfizer is positioning analytical data as a reusable asset that supports smarter science and faster innovation in pharmaceutical R&D.

