March 26, 2026
by Baljit Bains, Marketing Communications Specialist, ACD/Labs
ISO10993:18-2020—Simplifying the Chemical Characterization of Medical Device Materials
The presence of extractables and leachables (E&Ls) in medical devices after the manufacturing, packaging, or storage process is unavoidable. E&L analysis is integral to safeguard the safety of medical products and addresses the fundamental questions “What substance(s) may be released from drug packaging or medical devices?” and “Will it cause harm to patients?”
E&L studies require careful design and risk management and must be scientifically justified, considering factors such as device class, application of the device, and contact time. So, it is not surprising that in recent years E&L investigations are receiving increased attention from regulatory agencies.
Regulations govern everything from pre-market approvals and device listing to labeling, reporting, electronic signatures, and more. While there are many regulatory guidelines for medical devices, they can be ambiguous. This leaves scientists uncertain about whether they have fulfilled all the necessary regulatory requirements.

What are the ISO10993 Guidelines?
When it comes to medical devices—whether it’s a pre-filled syringe, skin patch, or implant—there is rarely only one applicable guideline. Most products need to meet multiple regulatory requirements, often overlapping, depending on how and where the device is used.
One of the key resources for manufacturers, suppliers, and labs is the ISO10993 series. These guidelines focus on the biological evaluation of medical devices and apply to those that come into direct or indirect contact with the human body and help ensure materials are safe for patient use.
ISO10993-1 frames biological evaluation as part of a broader risk management process. The latest version, ISO10993-1:2025, aligns more closely with ISO14971. It promotes and reinforces a structured approach to identify, assess, and manage biological risks based on the intended use of a device.
What are the ISO10993-18:2020 Guidelines?
Here, we focus on ISO10993-18:2020. This updated standard replaces the 2005 version. It includes improved flowcharts for the chemical characterization process, and offers clearer, more comprehensive definitions to enhance the understanding of analytical testing.
Core elements of ISO10993-18:2020 include determining E&Ls under real-use clinical conditions and using that information to assess biocompatibility.
Chemical Characterization of E&Ls
The ISO10993-18:2020 is a set of standards for characterizing medical devices. The guidelines are broken down into sections, starting with the chemical characterization procedure.
The setup of a chemical characterization study can be summarized in three steps—extraction, detection, and identification and quantification.
Extraction
Extraction aims to test the possible impact and the materials of medical devices. The ISO10993-18:2020 standards guide the selection of the most appropriate extraction conditions.
Selection of the extraction conditions depends on the type of medical device and its specific application. Although it may be necessary to use harsh extraction conditions, there is a fine balance that must be maintained. It is important to not select conditions that are too aggressive and alter the extractable profile.
The types of extraction include:
- Exaggerated Conditions—conditions that are intended to release a greater amount of chemical constituents compared to clinical conditions
- Exhaustive Extraction—multi-step extraction conducted until the amount of material extracted in a single extraction step is less than 10% of that determined in the initial extraction step
- Simulated Use Conditions—extraction using a method that simulates clinical use
Once the extraction type has been selected, other conditions like the extraction solvents, temperature, and time can also be decided.
Detection
The ISO10993-18:2020 guidelines provide recommendations to help select the most appropriate technique for analysis. Depending on the compound, the techniques required to analyze these compounds vary. The figure below shows the technique recommended for a specific category of compound.

Other factors to consider include:
- Application of the Analytical Technique—how will the technique be applied?
- Screening Approach—screen for every potential compound present in the extract
- Targeted Approach—detect what is known with a low limit of quantification (LOQ) and high accuracy
- Compound Class—to cover all classes of compounds (from volatile to non-volatile) multiple analytical techniques may need to be used
- Ionizable Species—it may be necessary to use high-sensitivity instruments to detect poorly ionized compounds
Considering all the factors mentioned above, the most appropriate analytical technique can be selected and applied, ensuring thorough extraction and detection of all compounds.
A Software Solution for Identification and Quantitation of E&Ls
Compounds detected at or above their toxicological thresholds are targeted for identification and quantitation.
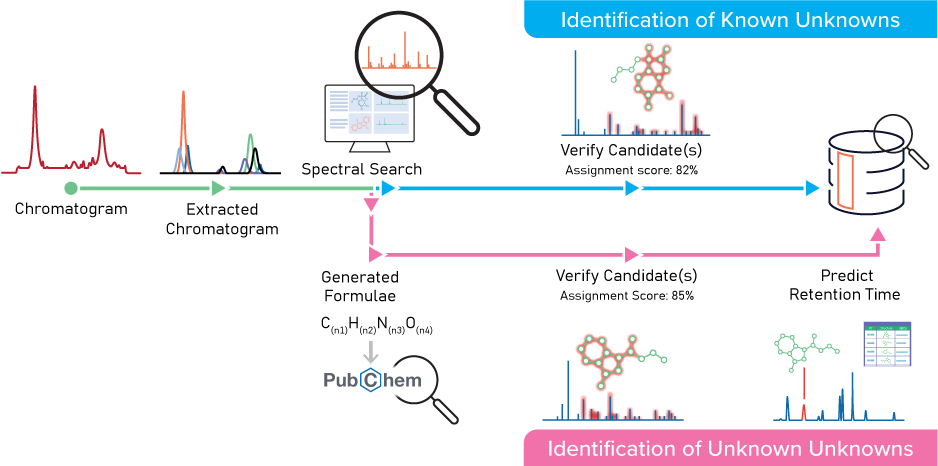
Software like ACD/Labs’ MS Structure ID Suite uses analytical chemistry to simplify, automate and streamline the chemical characterization of E&Ls in medical devices. It enables scientists to quickly identify ions present in the spectra, assign fragments, and verify results with confidence. All findings are captured and stored in a centralized database for easy access and traceability.
The workflow below is an example of how Medtronic uses MS Structure ID Suite to accelerate the chemical characterization of E&Ls. For more details, read the application note.
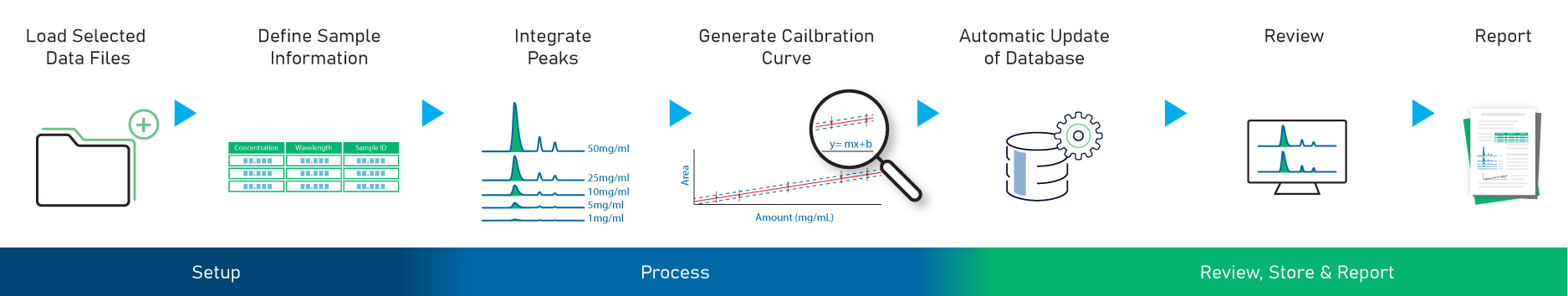
Once the analyte has been identified, the next step is to determine “how much”. Scientists can generate a calibration curve and quantitate the analytes. The quantitation workflow available in MS Structure ID Suite is shown in the image below. More details about the quantitation workflow can be found here.
MS Structure ID Suite can help accelerate component identification, raise the confidence level of identifications, and improve the accuracy of quantitation—ensuring the safety of your medical device.
Identification Status
ISO 10993-18:2020 requires the identification status of E&Ls to be indicated in chemical characterization reports. However, it is not possible to confirm every single identification with authentic reference sources. To sufficiently identify and report E&Ls it is necessary to establish and use levels of confidence. From high to low confidence, proposed levels are: confirmed, confident, tentative, speculative, and unidentified. The higher the confidence level, the lower the uncertainty to be used in toxicological assessment.
The highest level of confidence (confirmed identification) can only be used when 3 criteria are met:
- A sufficiently high match factor with a spectrum in a library
- Confirmation of the retention time of the compound on the used instrument
- The use of authentic standards to record the retention time and mass spectrum
It is especially important to have confirmed identifications when the reported concentration is close to the permitted daily dose of the compound.
Determining the Analytical Evaluation Threshold and Assessing Toxicological Risk
For all products on the market, it is necessary to conduct and submit toxicological risk assessments (TRA) to regulatory authorities. Allowable limits and AET must be considered when conducting a TRA. The process must also conform with other ISO guidelines (ISO10993-17, in particular).
There are detailed recommendations and guidelines within the ISO10993-18:2020 framework to help determine the AET. The AET is calculated based on the dosage and the route of administration, and acts as the baseline when a chemical poses a toxicological risk.
The threshold is set specifically for each chemical based on how the device is used, the type of contact and duration. Any compounds detected at or above this threshold must be identified, quantitated, and reported for toxicological assessment. This threshold is calculated using a dose-based threshold (DBT), along with the extraction volume, and the number of devices tested.
Depending on the chemical, the DBT is determined by either:
- Safety Concern Threshold (SCT)—below which a leachable has a dose so low that it presents negligible safety concerns from carcinogenic or non-carcinogenic toxic effects
- Threshold of Toxicological Concern—level of exposure of constituents, below which the risk for human health is not serious
Setting and meeting the AET is an important part of the regulatory submission process and also necessary for the safety of medical devices.
Safety Evaluation of Medical Devices
The evaluation of medical devices requires both chemical characterization and toxicology. Chemical characterization helps detect and measure compounds at very low levels. Toxicology then determines when those compounds could pose a risk to patients.
ISO10993-18:2020 brings these elements together. It offers clearer guidance on analytical testing and better aligns with other ISO guidelines.
Find out more about how AstraZeneca manages their E&Ls data, here.


