December 1, 2011
by Mikhail Elyashberg, Leading Researcher, ACD/Labs
For more than a decade, ACD/Structure Elucidator has been used by industry and academic experts to help solve some of the toughest structure problems. Using data from various analytical techniques (NMR, MS, UV, and IR) Structure Elucidator can propose chemical structures that are consistent with ALL available analytical data. The computer-aided structure elucidation (CASE) software has been used time and time again to help solve unknown structures and to correct erroneous proposed structures. Read on to see our featured story highlighting the use of Structure Elucidator and be sure to return to this page every month where we will highlight a new compound of interest.
The Initial Challenge
In a 2011 publication, Kummerlöwe and co-workers1 investigated one of the products obtained by reacting an azide-containing 1,5-enyne in the presence of electrophilic iodine sources.
After thorough collection, analysis, and interpretation of a comprehensive collection of analytical data, the authors concluded that they were unable to elucidate the chemical structure of the unknown by classical spectroscopic means.
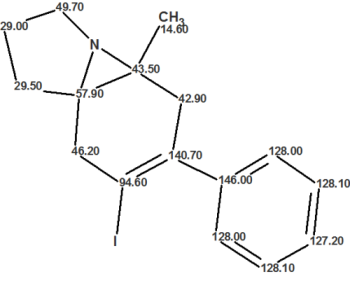
As a result, they proceeded with an attempt to solve the problem in an unconventional way by using Residual Dipolar Couplings (RDCs). The authors suggested 14 structures and selected the preferable one using RDC. The proposed structure based on RDC analysis is shown below:
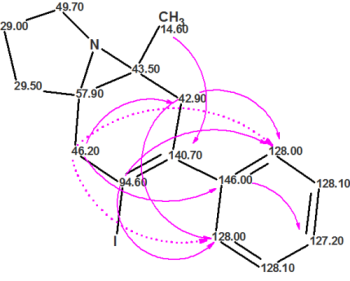
This structure was confirmed by a combination of 2D INADEQUATE data and 15N labeling. Further analysis revealed 9 non-standard carbon-carbon connectivities represented in the figure above with solid pink arrows. In addition, two unusually long and unexpected 5 bond heteronuclear correlations (denoted with the dashed pink arrows) were observed in the HMBC spectrum. Based on these nonstandard correlations, contradictory 2D NMR data was the main cause preventing structure elucidation using a traditional approach.
The Evidence
- The molecular formula for the unknown was C16H18IN
- The following spectroscopic data were acquired:
- IR spectrum
- 1D 1H NMR and 13C NMR
- 2D 1H-1H COSY, 1H-13C HSQC, 1H-13C HMBC (63 correlations), 1H-15N HMBC (7 correlations) and 1,1-ADEQUATE (900 MHz)
The CASE (Computer Assisted Structure Elucidation) Investigation
- ACD/Structure Elucidator was applied to solve this problem.2
- The first run included the following data based on the initial set of 2D NMR data presented by Kummerlöwe et al. 1
- 1D 13C
- 2D HSQC, 1H-13C HMBC, and 1H-15N HMBC (1,1-ADEQUATE was neglected)
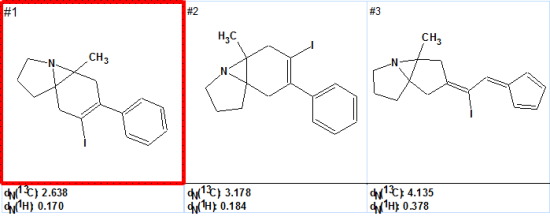
- The software generated 3 possible structures in 13 minutes.
- Following 1H and 13C chemical shift prediction, the structures were ranked in the following order with the chemical structure proposed in the original publication in the first position with the lowest chemical shift deviations. dN represent the chemical shift deviations (in ppm) between the assigned chemical shifts in the experimental spectrum and those generated by the ACD/Labs neural network prediction algorithm. The structure with the smallest deviation represents the best structure given the data.
- A second run was initiated with all of the data used above, plus the 1,1 ADEQUATE data.
- With the addition of this carbon-carbon connectivity information, the software generated only the correct structure in 0.7 seconds.
Conclusions
In this example, the authors were unable to elucidate the structure with traditional means, due to extremely contradictory 2D NMR data. The authors were able to determine the correct structure from a list of candidates based on the Residual Dipolar Couplings. Using a CASE system has the added benefit of automatically generating all possible structural candidates, and as such Structure Elucidator was able to unambiguously generate the correct structure immediately when given all the available data.
References
- G. Kummerlöwe, B. Crone, M. Kretschmer, S. F. Kirsch, B. Luy, Angew Chem Int Ed Engl, 2011, 50, 2643.
- Kirill Blinov, Mikhail Elyashberg, Ryan Sasaki, Sergey Molodstov, and Antony Williams, Poster at SMASH 2011, Chamonix, France.


